BEAM Reports Smaller-Than-Anticipated Q4 Loss, Year-Over-Year Revenue Growth
Beam Therapeutics Reports Improved Q4 2025 Results
Beam Therapeutics (BEAM) posted a fourth-quarter 2025 loss of $0.10 per share, excluding gains from equity investments. This result was significantly better than the consensus estimate, which anticipated a loss of $1.13 per share. In the same period last year, the company reported a loss of $1.09 per share.
Revenue for the quarter reached $114.1 million, far surpassing the expected $15 million and up from $30.1 million in the previous year’s fourth quarter. Most of this revenue came from licensing and collaboration agreements.
Over the past year, Beam Therapeutics’ stock price has increased by 8.6%, while the broader medical, biomedical, and genetics industry saw an 18% gain.
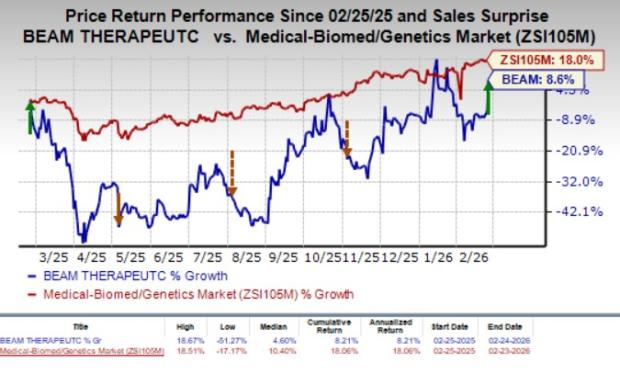
Source: Zacks Investment Research
Detailed Q4 Financials
- Research and development expenses for the quarter were $99.3 million, a 2.1% decrease compared to the same period last year.
- General and administrative costs rose by 12.6% year-over-year, totaling $32.3 million.
- As of December 31, 2025, Beam Therapeutics held $1.25 billion in cash, cash equivalents, and marketable securities, up from $1.1 billion at the end of September 2025.
Additionally, Beam Therapeutics secured a $500 million non-dilutive senior credit facility from Sixth Street to support the potential launch of ristoglogene autogetemcel (risto-cel) for sickle cell disease. The agreement includes an upfront payment of $100 million, up to $300 million tied to various milestones, and an optional $100 million tranche, all over a seven-year period. This financing is expected to extend the company’s cash runway into mid-2029.
Full-Year 2025 Performance
For the entire year, Beam Therapeutics generated $139.7 million in revenue, marking a 120% increase compared to the previous year.
Pipeline Progress and Clinical Updates
The company’s leading ex-vivo genome-editing therapy, risto-cel, is being evaluated in the phase I/II BEACON study for sickle cell disease (SCD). Updated results presented in December 2025 continued to demonstrate risto-cel’s unique benefits for SCD patients. Beam Therapeutics aims to file for a biologics license application (BLA) for risto-cel by the end of 2026.
Beam is also advancing its pipeline with BEAM-301 and BEAM-302, targeting glycogen storage disease type 1a (GSD1a) and alpha-1 antitrypsin deficiency (AATD), respectively. The first cohort of the phase I/II trial for BEAM-301 in GSD1a patients in the U.S. has completed dosing, and enrollment for the second cohort is underway. Initial results are anticipated in 2026.
BEAM-302 is being studied in a phase I/II dose-escalation trial for AATD. The company has reached an agreement with the FDA on a potential accelerated approval pathway based on 12-month biomarker data. Approximately 50 additional patients will be enrolled at the optimal dose to support a future BLA submission.
In the ongoing phase I study of BEAM-103, an anti-CD117 monoclonal antibody for SCD, dosing is expected to finish in the first half of 2026.
Beam Therapeutics is also broadening its liver-targeted genetic disease portfolio with BEAM-304 for phenylketonuria (PKU), a rare metabolic disorder. The company plans to file an investigational new drug application with the FDA in 2026.
Stock Performance and Analyst Ratings
Currently, Beam Therapeutics holds a Zacks Rank #4 (Sell).
Other biotech companies with higher Zacks rankings include:
- Harmony Biosciences (HRMY) – Zacks Rank #1 (Strong Buy)
- Assertio Holdings (ASRT) – Zacks Rank #1 (Strong Buy)
- Castle Biosciences (CSTL) – Zacks Rank #1 (Strong Buy)
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Why the Incoming Fed Chair Could Face Challenges in Reducing the Size of the Central Bank
Warner Bros. Discovery enters into a merger deal with Paramount Skydance

Dominion Energy's $890M Volume Surges to Rank 189 as Analysts Adjust Ratings and Capital Plan Expands
