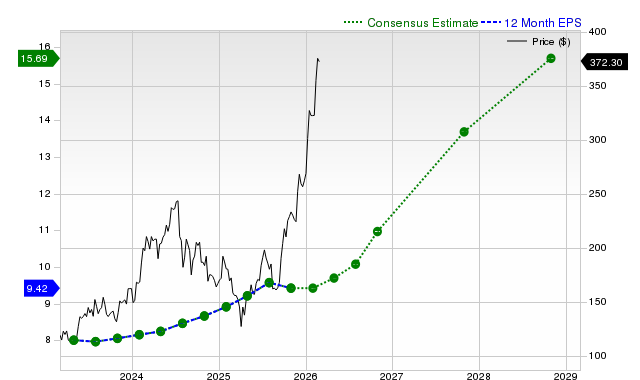
Larimar Secures $75M: Evaluating Financial Structure and Risk Premium Prior to a Major Catalyst
Overview of the $75 Million Capital Raise
Larimar has initiated a $75 million capital raise through an underwritten public offering of 18.75 million shares of common stock. Deerfield Management, the company’s largest shareholder, is leading the transaction and has agreed to acquire half of the shares at the public offering price. This strong institutional support serves as a foundation for the raise, reflecting ongoing confidence from a key long-term investor. The offering, conducted under a shelf registration effective since May 2024, enables Larimar to swiftly secure the funds necessary for advancing its lead asset, nomlabofusp.
Strategic Capital Deployment and Funding Path
This capital raise follows a consistent pattern, coming on the heels of a $69 million raise in July 2025. The company’s disciplined approach aims to finance the clinical development of nomlabofusp and provide working capital, with a direct focus on supporting a pivotal Biologics License Application (BLA) submission. For institutional investors, this represents a classic allocation of capital toward a high-impact, binary event with the potential for significant returns.
Risks and Market Dynamics
Despite the strategic rationale, this transaction brings notable short-term risks. The offering results in considerable dilution, and the stock’s recent volatility highlights heightened speculative interest ahead of the BLA milestone. Over the past five days, shares have surged 106%, with a turnover rate of 167%, indicating intense trading activity as investors position for the upcoming catalyst. Such liquidity dynamics make the stock highly reactive to any developments related to the BLA timeline or clinical data. Investors must construct their portfolios with the binary nature of the event in mind, preparing for potential turbulence as the catalyst approaches.
The Key Catalyst: FDA Breakthrough Therapy and June 2026 BLA Submission
The investment case centers on a binary outcome, with the company’s near-term prospects hinging on the successful execution of its regulatory strategy. The main catalyst is the anticipated BLA submission in June 2026 for nomlabofusp targeting Friedreich's Ataxia (FA). This timeline is crucial for justifying the recent capital raise, as it defines the window for a potentially transformative event.
Larimar’s regulatory position is robust, bolstered by the FDA’s recent Breakthrough Therapy Designation for nomlabofusp, which was granted based on early clinical evidence of meaningful improvement. Notably, 100% of participants in an open-label study achieved skin FXN levels comparable to asymptomatic carriers after six months—a key surrogate endpoint for accelerated approval. Topline data from this study, expected in Q2 2026, will directly inform the BLA submission. The company plans to follow with a confirmatory Phase 3 trial, targeting a potential U.S. launch in the first half of 2027 if approval is granted.
This scenario creates a high-conviction opportunity for portfolio allocation. The capital raise eliminates a near-term funding risk, but the binary nature of the outcome remains paramount. The result of the June 2026 BLA submission will determine the company’s valuation: success could lead to a dramatic re-rating, while any negative outcome—whether due to data, safety, or regulatory concerns—could result in a sharp decline. The recent surge in share price reflects the market’s anticipation of this pivotal event.
Given the stock’s volatility and liquidity, this is a high-risk, short-duration investment. Institutional investors should size their positions according to their risk tolerance for concentrated, event-driven bets. The opportunity is not about gradual growth, but about capturing a potential inflection point, requiring a portfolio approach that acknowledges the all-or-nothing nature of the catalyst.
Financial Implications and Portfolio Considerations
This offering significantly dilutes existing shareholders, increasing the share count by roughly 50% compared to the July 2025 raise. The company expects to generate $56.4 million in net proceeds from the sale, which will fund the BLA submission and the Phase 3 trial. Combined with a $175.4 million cash position as of September 2025, this capital is projected to extend the company’s runway into the fourth quarter of 2026. While this buys time, it comes at the cost of a much larger equity base.
The stock’s recent performance—marked by a 106% five-day gain and extreme volatility—underscores its speculative nature. This is not a play on quality fundamentals, but a high-beta, event-driven wager. Portfolio allocation should reflect the concentrated risk, treating the position as a single-asset bet rather than a diversified holding. The risk premium is already reflected in the stock’s price swings, and any setback with the June 2026 BLA could trigger a sharp selloff.
For institutional investors, the risk/reward profile is asymmetric and highly concentrated. The capital raise funds the final stretch toward a potentially transformative event, but does not change the binary outcome. Position sizing should be conservative, given the volatility and the all-or-nothing nature of the catalyst. This is a speculative allocation, with returns tied to a single clinical milestone and risk defined by the stock’s liquidity and price fluctuations.
Key Catalysts, Risks, and Monitoring Points
Institutional investors should focus on a well-defined set of upcoming events and risks. The primary catalyst is the topline data from the open-label study expected in Q2 2026, which will underpin the planned BLA submission in June 2026. This data release will be the main validation for the investment thesis. Achieving consistent clinical benefit and meeting the FDA’s surrogate endpoint for accelerated approval could set the stage for a major re-rating, while any delays or disappointing results could lead to a significant decline.
A critical risk to monitor is the safety profile, particularly regarding anaphylaxis. Early trial results showed 7 cases of anaphylaxis among 39 participants during the initial six weeks of treatment. Although these events resolved with standard care and were more common in those with prior exposure, this safety signal will be closely examined by regulators and payers. The FDA has indicated that the adequacy of the safety database will be a key focus at the time of BLA review. Investors should watch for updates on the implementation of a modified starting dose regimen and any feedback from the Phase 3 study regarding safety.
Progress on the confirmatory Phase 3 trial is another important milestone. The company is preparing to launch this global study, with enrollment already underway. The trial’s design and enrollment pace will be critical indicators of Larimar’s operational execution and its ability to support full approval following an accelerated launch. Any significant delays or operational challenges could jeopardize the targeted first-half 2027 launch.
In summary, the investment setup is high-conviction and binary. The key monitoring points are the Q2 2026 data readout, the June BLA submission, and ongoing management of the anaphylaxis safety risk. Portfolio construction should reflect the event-driven volatility, with clear plans for adjusting or exiting positions based on these specific milestones.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Is CLEAR Secure, Inc. (YOU) a Good Investment as a Trending Stock Right Now?
Here’s What You Should Understand About Zscaler, Inc. (ZS) Besides Its Popularity
Here’s What You Should Understand About Intuitive Surgical, Inc. (ISRG) Besides Its Popularity
Applied Materials, Inc. (AMAT) is Drawing Interest from Investors: What You Need to Understand