DNLI Q4 Reports Smaller-Than-Anticipated Loss, Spotlight on Hunter Syndrome Treatment
Denali Therapeutics Q4 2025 Financial Overview
Denali Therapeutics (DNLI) posted a fourth-quarter 2025 net loss of $0.73 per share, which was slightly better than the consensus estimate of a $0.75 loss. This compares to a loss of $0.67 per share in the same period last year.
The year-over-year increase in per-share loss was primarily driven by higher operating expenses.
Since Denali does not have any products on the market, its revenue is limited to proceeds from ongoing partnerships. In the reported quarter, the company did not record any collaboration revenue, falling short of the expected $18 million.
Over the past year, DNLI shares have risen by 13.4%, outperforming the medical, biomedical, and genetics sector, which saw an 11.5% increase.
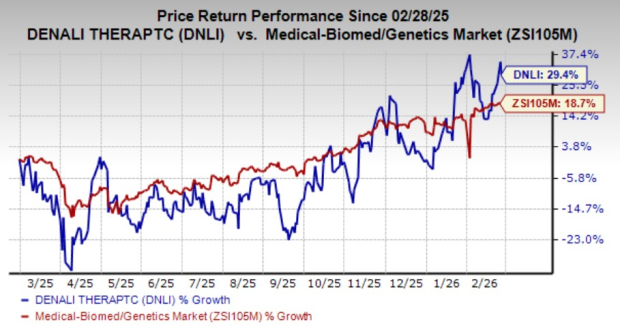
Source: Zacks Investment Research
Key Points from Q4 Results
- Research and development costs fell by 1.9% to $97.9 million, largely due to reduced spending on small molecule projects.
- General and administrative expenses climbed 31.3% to $39.5 million, reflecting preparations for the anticipated launch of tividenofusp alfa.
- As of December 31, 2025, Denali held approximately $966.2 million in cash, cash equivalents, and marketable securities.
2025 Full-Year Summary
- No revenue was generated during 2025.
- Annual loss per share widened to $2.97, compared to $2.57 in 2024.
Progress on Lead Drug Candidate
Denali is actively preparing for the commercial introduction of its lead asset, tividenofusp alfa.
The biologics license application (BLA) for tividenofusp alfa, aimed at treating mucopolysaccharidosis type II (Hunter syndrome), is currently under review by U.S. regulators.
The FDA extended its review deadline from January 5, 2026, to April 5, 2026.
Tividenofusp alfa has received several designations from the FDA, including Breakthrough Therapy, Fast Track, Orphan Drug, and Rare Pediatric Disease status.
This investigational therapy is designed to deliver the iduronate-2-sulfatase (IDS) enzyme across the blood-brain barrier, targeting both the body and brain.
The BLA review period was lengthened after Denali provided updated clinical pharmacology data in response to an FDA request. The company clarified that this update was not related to efficacy, safety, or biomarkers.
The FDA classified the submission as a major amendment, resulting in the extension of the action date.
The ongoing global phase II/III COMPASS trial is expected to provide supporting evidence for regulatory submissions worldwide, with enrollment for neuronopathic participants (Cohort A) completed in December 2025.
Pipeline Developments
- Denali is investigating DNL126 for Sanfilippo syndrome type A (MPS IIIA). Preliminary phase I/II results presented at WORLDSymposium showed significant reductions in key disease biomarkers and a safety profile similar to existing enzyme replacement therapies. These findings may support an accelerated approval pathway, and a phase III confirmatory study is planned.
- Other pipeline candidates are being developed in collaboration with Takeda (TAK), Biogen (BIIB), and Sanofi (SNY).
- Denali and Takeda are advancing DNL593, a potential treatment for granulin mutation-associated frontotemporal dementia (FTD-GRN). Enrollment in the phase I/II study is complete, with 40 participants. Initial patient data is expected in 2026.
- Denali recently received approval for a phase Ib trial of DNL628, with preparations underway.
- Denali and Biogen are jointly developing BIIB122. Biogen is leading the global phase IIb LUMA study for early-stage Parkinson’s disease, with results anticipated mid-2026. Denali is running the phase IIa BEACON study to evaluate LRRK2 inhibition in LRRK2-associated Parkinson’s disease.
- In October, Denali submitted an IND for DNL952 (ETV:GAA) to begin clinical studies in Pompe disease. The FDA has since lifted the clinical hold, and phase I study preparations are ongoing.
- Sanofi is developing eclitasertib for moderate to severe ulcerative colitis, with phase II data expected in the first half of the year.
Analysis of Denali’s Outlook
Approval of tividenofusp alfa could significantly enhance Denali’s growth trajectory. Progress with DNL126 is also promising.
The company’s robust cash reserves highlight its capacity to support ongoing research and development initiatives.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
All about U.S Congress’s new bill and its intent to protect open-source developers

Southwest's Rally: A Guidance Reset or a Priced-In Beat?

TERN-701 ASH Data: Is This a Strategic Opportunity for Revaluation or Has the Market Already Factored It In?
