Kymera Q4 Loss Exceeds Projections, Additional Funding Prolongs Financial Runway
Kymera Therapeutics Q4 2025 Financial Overview
Kymera Therapeutics (KYMR) posted a net loss of $0.97 per share for the fourth quarter of 2025, exceeding analysts’ expectations, which had forecasted a loss of $0.77 per share. This result was also worse than the loss of $0.88 per share reported in the same period last year. The increased deficit was mainly attributed to higher research and development costs and a decline in collaboration revenue.
Collaboration revenue for the quarter reached $2.9 million, falling short of the consensus estimate of $30 million. These earnings were primarily generated from Kymera’s partnership with Gilead Sciences (GILD).
In the previous year’s fourth quarter, Kymera earned $7.4 million in collaboration revenue through its alliance with Sanofi (SNY).
Over the past year, Kymera’s stock price has surged by 214.2%, significantly outpacing the broader industry’s growth of 18.6%.
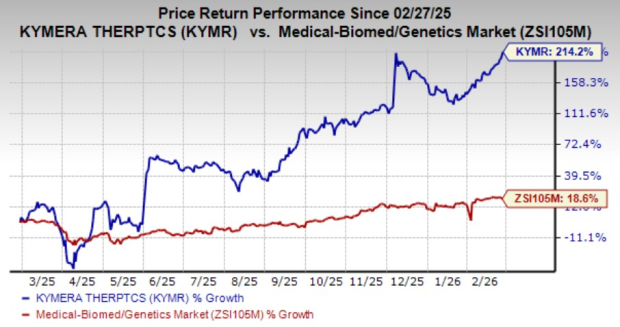
Image Source: Zacks Investment Research
Rising Operating Expenses in Q4
Research and development expenditures climbed to $83.8 million, marking a 16.7% increase from the prior year. This rise was largely due to expanded investments in the STAT6 program, platform development, discovery initiatives, and the continued expansion of the R&D team.
General and administrative costs grew by 3.7% year-over-year, reaching $16.9 million. The increase was driven by higher legal and professional service fees, as well as greater personnel and facility expenses.
In December 2025, Kymera completed a public equity offering, raising approximately $692 million in gross proceeds, including the full exercise of the overallotment option. This financing boosted the company’s cash reserves to about $1.6 billion at year-end and extended its financial runway through 2029.
2025 Annual Results
Kymera’s total revenue for 2025 dropped by 16.8% to $39.2 million, missing the consensus estimate of $54.7 million.
The company’s loss per share widened to $3.69, compared to $2.98 in 2024.
Pipeline Developments
Kymera is advancing targeted protein degradation (TPD), a cutting-edge therapeutic approach that leverages the body’s natural cellular recycling mechanisms to selectively remove harmful proteins.
- STAT6 Degrader Program: KT-621, a first-in-class oral STAT6 degrader, is in phase 2 trials for atopic dermatitis and asthma. It targets the main driver of type II inflammation. In December 2025, positive phase Ib BroADen study results demonstrated robust STAT6 degradation, significant biomarker reductions, and notable clinical improvements with a favorable safety profile.
- In January 2026, the BROADEN2 phase IIb study for atopic dermatitis was expanded to include adolescents, and dosing began in the BREADTH study for eosinophilic asthma. Both global, placebo-controlled trials are underway, with results expected between mid and late 2027.
- KT-621 received Fast Track designation for moderate-to-severe atopic dermatitis in December 2025.
- IRF5 Degrader Program: KT-579, an oral IRF5 degrader, is in phase 1 development. It aims to suppress pro-inflammatory cytokines, Type I interferons, and autoantibody production while maintaining normal immune function. Following FDA IND clearance, dosing began in a phase I study with healthy volunteers, and data is anticipated in the second half of 2026.
- Partnered Programs: KT-485/SAR447971, a selective oral IRAK4 degrader, is being co-developed with Sanofi for immuno-inflammatory diseases. IND-enabling studies are complete, and clinical trials are expected to start in 2026.
- Preclinical research is ongoing with Gilead Sciences under an exclusive option and license agreement to advance Kymera’s oral CDK2 molecular glue program for breast cancer and other solid tumors. If Gilead exercises its option, it will pay a fee and take over development, manufacturing, and commercialization.
Assessment of Kymera’s Performance
Kymera’s research and development spending continues to rise as its pipeline progresses. The company remains financially strong, ending 2025 with roughly $1.6 billion in cash and sufficient funding to support operations through 2029. Its innovative TPD platform and ongoing pipeline advancements are promising.
The investment outlook is focused on successful execution in targeted protein degradation, especially with KT-621’s phase 2 trials for atopic dermatitis and asthma. Further positive updates on KT-621 could drive additional gains for the stock.
Kymera’s Zacks Rank and Alternative Biotech Picks
Kymera Therapeutics currently holds a Zacks Rank #3 (Hold). Castle Biosciences (CSTL), with a Zacks Rank #1 (Strong Buy), is a higher-rated alternative.
- Castle Biosciences’ projected 2026 loss per share has improved from $1.06 to $0.96 over the past 60 days.
- CSTL shares have advanced 26.2% in the last six months.
- Castle Biosciences exceeded earnings estimates in three of the past four quarters, with an average surprise of 66.11%.
Zacks Research: Top Stock Picks
The Zacks research team has identified five stocks with the highest potential to double in value in the coming months. Among these, Director of Research Sheraz Mian highlights a satellite-based communications company poised for rapid growth. The space industry is expected to reach a trillion-dollar valuation, and this firm’s expanding customer base and projected revenue surge in 2025 make it a standout. While not all picks achieve such gains, this one could outperform previous Zacks selections like Hims & Hers Health, which soared by over 200%.
Additional Free Stock Analysis Reports
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Saudi and Egyptian stock exchanges tumble amid regional turmoil caused by US-Iran tensions
Hyperliquid price prediction – HYPE eyes $38, but watch THIS golden pocket first
If You’re Bearish on XRP, Listen to What David Schwartz Said
刚刚!央行重磅表态:降准还有一定空间
