Deucrictibant Data Supporting Potentially Differentiated Profile for the On-Demand and Prophylactic Treatment of Bradykinin-Mediated Angioedema Presented at AAAAI 2026
- RAPIDe-3 met the primary and all 11 secondary efficacy endpoints with high statistical significance with deucrictibant achieving onset of symptom relief in 1.28 hours and complete symptom resolution in 11.95 hours, and confirming its potentially differentiated profile for the treatment of HAE attacks
- Final CHAPTER-1 data provide further evidence on long-term safety and efficacy of deucrictibant for the prevention of HAE attacks; single-dose, sustained therapeutic exposure with deucrictibant extended-release tablet supports once-daily prophylactic application
ZUG, Switzerland, March 02, 2026 (GLOBE NEWSWIRE) -- Pharvaris (Nasdaq: PHVS), a late-stage biopharmaceutical company developing novel, oral bradykinin B2 receptor antagonists to help address unmet needs of those living with bradykinin-mediated diseases such as hereditary angioedema (HAE) and acquired angioedema due to C1 inhibitor deficiency (AAE-C1INH), summarized the presentations for the American Academy of Allergy, Asthma & Immunology (AAAAI) 2026 Annual Meeting, which took place from February 27-March 2, 2026, in Philadelphia, PA.
“The RAPIDe-3 data confirm the robust and consistent clinical effects of deucrictibant across our primary and all 11 secondary efficacy endpoints versus placebo, confirming its potentially differentiated profile for the treatment of all types of HAE attacks,” said Peng Lu, M.D., Ph.D., Chief Medical Officer of Pharvaris. “Due to its mechanism of action, deucrictibant is expected to outcompete bradykinin at the B2 receptor, resulting in direct modulation of bradykinin signaling, as demonstrated by the rapid and sustained symptom relief and attack resolution findings from RAPIDe-3. We are pleased to present these pivotal data at AAAAI and look forward to future exchanges with the HAE community about deucrictibant’s potential to become standard of care in the treatment of HAE attacks.”
Details of the presentations are outlined below:
On-Demand Therapy
Oral Deucrictibant Immediate-Release Capsule in Treatment of Hereditary Angioedema Attacks: Results of the Phase 3 RAPIDe-3 Study, presented by Marc A. Riedl, M.D., M.S. in a featured poster. The RAPIDe-3 (NCT06343779) global Phase 3, placebo-controlled study evaluated orally administered deucrictibant immediate-release capsule (20 mg) for the on-demand treatment of attacks in participants 12 years and older with HAE, including those with HAE with normal C1 inhibitor. Final results from RAPIDe-3 provide further evidence on the rapid and sustained efficacy, safety, and tolerability of deucrictibant. Results from this study met the primary and all 11 secondary efficacy endpoints. The median time to onset of symptom relief, the primary endpoint, was 1.28 hours with deucrictibant treatment versus over 12 hours with placebo. This endpoint was achieved by 12 hours in 90.4% of deucrictibant-treated attacks, versus 48.3% of placebo-treated attacks. The median time to End of Progression™ of attack symptoms was 17.47 minutes with deucrictibant treatment versus 228.67 minutes with placebo. The median time to complete resolution of attack symptoms was 11.95 hours with deucrictibant treatment versus over 48 hours with placebo. This endpoint was achieved by 48 hours in 81.9% of deucrictibant-treated attacks, versus 36.8% of placebo-treated attacks. Deucrictibant was well tolerated with no treatment-related serious adverse events and no participants discontinuing treatment due to treatment-emergent adverse events.
Content Validity of the Angioedema symptom Rating scAle (AMRA) to Assess Symptoms of Hereditary Angioedema Attacks, presented by Teresa Caballero, M.D., Ph.D. A collated analyses from a mixed-methods study and RAPIDe-3 provided confirmatory evidence to support the validity of AMRA-3 and AMRA-5 in assessing the severity of key symptoms associated with HAE attacks. Participants across age groups, including adolescents and adults, reported a variety of symptoms when experiencing HAE attacks, with the most frequently observed symptoms captured by the AMRA-3 scale or, for people experiencing upper airway attacks, including laryngeal attacks, by the AMRA-5 scale.
Long-Term Prophylaxis
Long-Term Safety and Efficacy of Oral Deucrictibant for Prophylaxis in Hereditary Angioedema: Final Results of the Phase 2 CHAPTER-1 Open-Label Extension Study, presented by John Anderson, M.D. in a featured poster. Data from the final analysis of the open-label extension (OLE) of the two-part Phase 2 clinical study of deucrictibant for the long-term prophylaxis of HAE attacks, CHAPTER-1, provided further evidence about deucrictibant’s profile being well tolerated with no safety signals observed in the OLE for up to approximately three years; mean systolic and diastolic blood pressure remained stable throughout all study assessments. The attack rate reduced within one week of deucrictibant treatment remained low for up to approximately three years: the mean attack rate was reduced from a study baseline of 2.18 attacks/month to 0.12 attacks/month in the open-label extension. Additionally, approximately half of the participants in the open-label extension were attack free.
Long-Term Prophylactic Treatment With Oral Deucrictibant Improved Health-Related Quality of Life in Participants With Hereditary Angioedema: Final Results of the Phase 2 CHAPTER-1 Open-Label Extension Study, presented by Michael E. Manning, M.D. Data from the final analysis of the open-label extension (OLE) of the two-part Phase 2 clinical study of deucrictibant for the long-term prophylaxis of HAE attacks, CHAPTER-1, showed that treatment with deucrictibant resulted in clinically-meaningful improvements in health-related quality of life (HRQoL) and disease control and in higher treatment satisfaction for up to approximately three years.
Sustained Therapeutic Exposure with Once-Daily Oral Deucrictibant Extended-Release Tablet for Prophylaxis of Hereditary Angioedema Attacks, presented by Zhi-Yi Zhang, Ph.D. in a featured poster. Phase 1 data supporting the once-daily applicability of deucrictibant extended-release tablet include its single-dose pharmacokinetic profile under fasted and fed conditions and the sustained (≥24 hours) therapeutic exposure during repeat dosing.
Beyond HAE
A Novel Kinin Biomarker Assay for Characterization of Different Types of Bradykinin-Mediated Angioedema, presented by Evangelia Pardali, Ph.D. Pharvaris has developed an assay that can measure the levels of bradykinin and other kinin related peptides in plasma to characterize people with bradykinin-mediated angioedema. In addition to clearly showing bradykinin-forming cascade sensitivity in people with multiple types of HAE and with AAE-C1INH, cold activation caused increased bradykinin levels in samples from individuals with HAE with normal C1 inhibitor of unknown aetiology and angioedema of unknown cause, indication that the angioedema attacks in these individuals may be bradykinin-mediated. The clinically validated kinin biomarker assay may become a key tool for identifying, studying, and managing bradykinin-mediated diseases, including bradykinin-mediated angioedema.
About Deucrictibant
Deucrictibant is a novel, potent, orally bioavailable small-molecule bradykinin B2 receptor antagonist currently in clinical development. Deucrictibant is being investigated for its potential to prevent the occurrence of bradykinin-mediated angioedema attacks and to treat the manifestations of attacks if/when they occur by inhibiting bradykinin signaling through the bradykinin B2 receptor. Pharvaris is developing two formulations of deucrictibant for oral administration: an extended-release tablet to enable sustained absorption and efficacy as prophylactic treatment, and an immediate-release capsule to enable rapid onset of activity for on-demand treatment. Deucrictibant has been granted orphan drug designation for the treatment of bradykinin-mediated angioedema by the U.S. Food and Drug Administration, the European Commission, and Swissmedic.
About Pharvaris
Pharvaris is a late-stage biopharmaceutical company developing novel, oral bradykinin B2 receptor antagonists to help address unmet needs in bradykinin-mediated conditions, including all types of bradykinin-mediated angioedema. Pharvaris’ aspiration is to offer therapies with injectable-like efficacy™, a well-tolerated profile, and the convenience of oral administration to prevent and treat bradykinin-mediated angioedema attacks. By delivering on this aspiration, Pharvaris aims to provide a new standard of care in bradykinin-mediated angioedema. Pharvaris is preparing marketing authorization applications for deucrictibant immediate-release capsule as an on-demand treatment of HAE attacks, and a global pivotal Phase 3 study of deucrictibant extended-release tablet for the prevention of HAE attacks (CHAPTER-3) is ongoing with topline data anticipated in the third quarter of 2026. In addition, CREAATE is an ongoing Phase 3 study of deucrictibant for the prophylactic and on-demand treatment of AAE-C1INH attacks. For more information, visit .
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
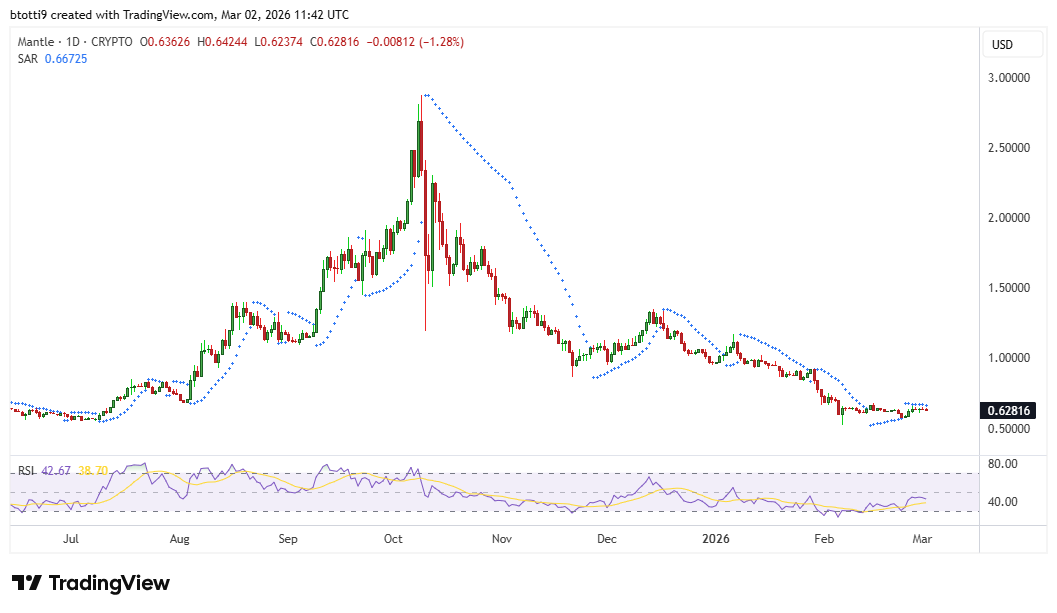
Mantle hits $1B market size milestone on Aave: will MNT price explode next?
Block Street Launches BSB Token to Bridge Crypto Infrastructure Gaps
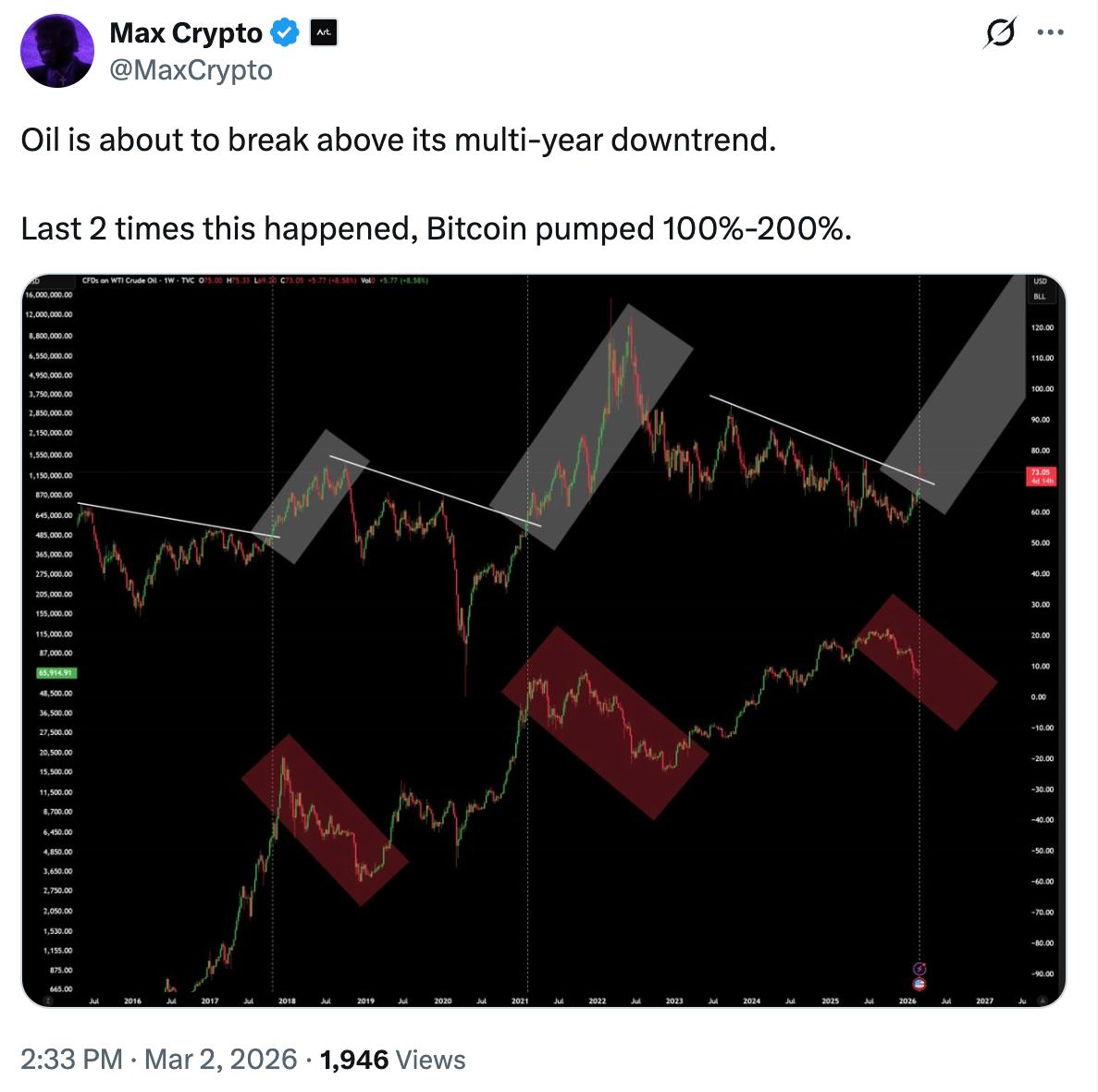
Will Bitcoin crash if oil prices hit $100 per barrel?