MRNA Shares Rise Following CHMP Approval of Combined COVID-19 and Flu Vaccine
Moderna's Shares Climb Following Positive CHMP Recommendation
Moderna's stock experienced a roughly 4% increase on Friday after the company revealed that the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) had recommended approval for mCombriax (mRNA-1083). This vaccine, which utilizes mRNA technology, is designed to protect against both COVID-19 and influenza.
Next Steps for mCombriax in the EU
The CHMP’s endorsement will now be evaluated by the European Commission, with a final verdict anticipated soon. Should mCombriax receive approval, it will become Moderna’s fourth product available in the European market, joining Spikevax, mNexspike (both COVID-19 vaccines), and the RSV vaccine mResvia.
Clinical Trial Results Support Approval
The CHMP’s positive opinion is based on results from a pivotal phase III clinical trial, which achieved its main objectives. Findings demonstrated that a single dose of mCombriax triggered stronger immune responses against both COVID-19 and flu compared to currently approved standalone vaccines for each disease.
This large-scale study involved approximately 8,000 adult participants, split into two age groups. The first group, adults aged 65 and above, received either mCombriax or a combination of Fluzone HD (a high-dose flu vaccine from Sanofi) and Spikevax. The second group, adults aged 50 to 64, compared mCombriax with Fluarix (a standard-dose flu vaccine from GSK) administered alongside Spikevax.
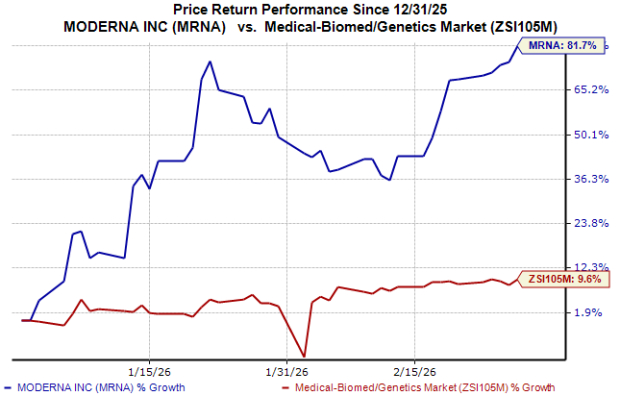
MRNA Stock Performance in 2024
Since the start of the year, Moderna’s share price has soared by about 82%, significantly outpacing the industry average growth of 10%.
Details on mCombriax and Regulatory Progress
mCombriax is a combination of Moderna’s mNexspike and mRNA-1010, an experimental influenza vaccine. Regulatory submissions are under review in the United States, Europe, Canada, and Australia, with a decision from the FDA expected by August 5, 2026.
Moderna initially filed for FDA approval of mCombriax, but withdrew the application in May 2025 after the agency requested more efficacy data for the flu component. The company has since provided the necessary data as part of the mRNA-1010 submission and is awaiting further feedback from the FDA regarding a potential resubmission for mCombriax.
Regulatory Challenges in the U.S.
Vaccine demand in the U.S. has been impacted by increased regulatory scrutiny. The FDA initially declined to review the application for mRNA-1010, citing concerns that the comparator vaccine used in the trial was not the optimal standard of care. However, after Moderna proposed a revised regulatory approach based on age groups, the FDA reconsidered. The updated submission now seeks full approval for adults aged 50-64 and accelerated approval for those 65 and older.
Moderna, Inc. Stock Information
For more details on Moderna’s stock price and performance, visit the official stock chart and quote pages.
Current Zacks Rank for Moderna
Moderna holds a Zacks Rank #3 (Hold).
Top 5 Stocks with High Growth Potential
- Stock #1: A disruptive company demonstrating strong growth and resilience
- Stock #2: Showing bullish indicators and potential for rebound
- Stock #3: Considered one of the most attractive investment opportunities
- Stock #4: Leading a rapidly expanding industry
- Stock #5: An innovative omni-channel platform ready for expansion
Many of these stocks are not yet widely recognized by Wall Street, offering early investment opportunities. Previous recommendations from this report have achieved gains of +171%, +209%, and +232%.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
AEHL Drops 18.8% Amid Intraday Turmoil: What’s Behind the Sharp Decline?
Why You Should Pay Close Attention to Zscaler (ZS) Global Revenue Patterns
Countdown to Marvell (MRVL) Q4 Earnings: Examining Projections Beyond Just Revenue and EPS
MasTec, Inc. (MTZ) Reaches New Peak: Can the Rally Continue?
