Hologic's Diagnostic Capabilities Spotlighted: Could There Be Further Growth?
Hologic Diagnostics Segment Overview
In the first quarter of fiscal 2026, Hologic (HOLX) reported $464.4 million in Diagnostics revenue, marking a 1.3% decrease compared to the previous year. The division continues to be driven by molecular diagnostic tests, which utilize the Panther and Panther Fusion platforms. However, the period saw a 3.5% drop in Molecular Diagnostics revenue, primarily due to reduced demand for COVID-19 and older STI tests.
Despite these challenges, increased sales of BV, CV/TV tests and Panther Fusion assays helped offset some losses. Hologic is focusing on expanding its presence in the U.S. vaginitis market by promoting its high-throughput solutions and improving reimbursement rates. The Panther Fusion system’s open access feature enables laboratories to run both lab-developed and in vitro diagnostic tests on a single device. Last year, the company achieved FDA 510(k) clearance and CE-IVDR certification for its Panther Fusion Gastrointestinal Bacterial and Expanded Bacterial Assays, supporting its strategy to broaden its test menu. Additionally, Biotheranostics has seen greater use of its Breast Cancer Index, which assists in determining extended endocrine therapy for patients.
Cytology and Perinatal Segment Performance
Revenue from Cytology and perinatal products increased by 1.1% during the quarter. Sales of ThinPrep Pap tests were negatively affected by geopolitical tensions between the U.S. and China, as well as regulatory changes in China, leading to lower revenue in the Asia-Pacific region. The Genius Digital Diagnostics System, which combines advanced AI technology with digital imaging to identify precancerous and cancerous cervical cells, is positioned as a potential growth catalyst.
Recent Regulatory Milestones
February 2026 brought positive developments for Hologic. The Aptima HPV Assay received FDA approval for clinician-collected HPV primary screening, further strengthening the company’s Cervical Health Portfolio. The Genius System also obtained expanded CE certification in the European Union, now authorized for imaging and reviewing both cellular and tissue samples.
Peer Company Updates: BD and Abbott
Becton, Dickinson and Company (BDX) recently secured CE Mark for its Revello Vascular Covered Stent, a new solution for treating atherosclerotic lesions in the common and external iliac arteries. This innovation comes as peripheral artery disease, particularly involving the iliac arteries, remains a significant health concern among Europe’s aging population.
Abbott (ABT) has received FDA approval for its CardioMEMS HERO, a next-generation pulmonary artery pressure monitoring device. The updated design aims to provide heart failure patients and their healthcare providers with daily insights to better manage disease progression. Abbott also earned CE Mark in Europe for the TactiFlex Duo Ablation Catheter, Sensor Enabled, which advances pulsed field ablation technology for atrial fibrillation treatment.
HOLX Stock Analysis and Outlook
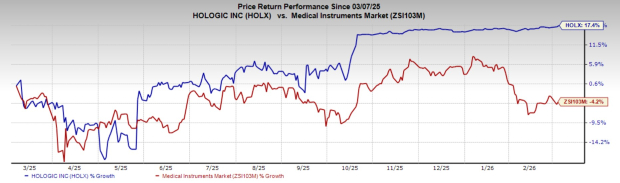
Over the past year, Hologic’s stock has climbed 17.4%, while the broader industry experienced a 4.2% decline.
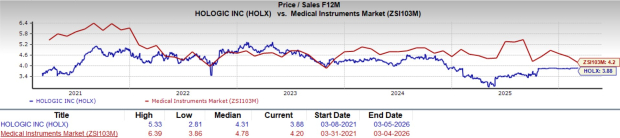
Currently, Hologic trades at a forward five-year price-to-sales ratio of 3.88X, which is below both its historical median and the industry average.
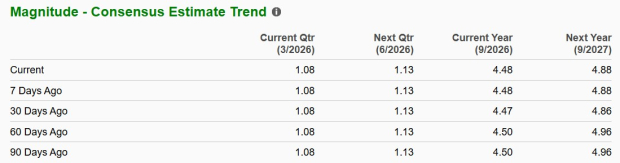
Below is a summary of Hologic’s recent earnings estimate trends.
At present, HOLX holds a Zacks Rank #4 (Sell).
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Lantronix’s MediaTek Edge AI Venture Awaits Proof at Embedded World 2026 Challenge
Pump.fun team moves 1.75B PUMP: Can bulls offset selling pressure?
Robinhood’s startup fund stumbles in NYSE debut

Robinhood’s startup fund stumbles in NYSE debut
