Second Study Affirms Superiority of iMDx GraftAssure Assay's Proprietary dd-cfDNA Combination Model Score
- New data supports GraftAssure commercialization efforts
- Multi-center study featured 249 biopsy-matched patients
- Proprietary “Combination Model” score outperformed percentage-only measures of dd-cfDNA
- Reinforces data presented at World Transplant Congress in 2025
NASHVILLE, Tenn., March 13, 2026 -- Insight Molecular Diagnostics Inc. (Nasdaq: IMDX) (“iMDx”) today highlighted the publication of a new peer-reviewed study demonstrating the potential superiority of the company’s flagship GraftAssure
The study, published in Transplant International, and titled “Donor-Derived Cell-Free DNA as a Non-Invasive Readout of Activity Across the Rejection Continuum,” was conducted by investigators from Heidelberg University Hospital and Charité – Universitätsmedizin Berlin and included iMDx scientist authors.
This new study, combined with data presented at World Transplant Congress in 2025, point to growing clinical differentiation and marketability of the GraftAssure technology and support iMDx’s strategic vision. Beginning in 2026, iMDx seeks to deliver the industry-leading molecular diagnostic test kit for clinical use that expands and improves access to organ health testing for kidney transplant patients. The company expects that enabling localized testing will deliver new value in the roughly $2 billion-plus addressable market for regulated transplant rejection testing.
The new peer-reviewed publication reports on 249 patients, who had undergone clinically indicated kidney transplant biopsies, from two independent cohorts, comparing dd-cfDNA measurements in blood plasma with newly developed histopathology-derived rejection indices
The investigators found that dd-cfDNA levels increased with histological evidence of graft inflammation, with the strongest associations observed in microvascular inflammation (MVI) and antibody-mediated rejection (AMR), which are the most clinically significant forms of transplant rejection.
IMDx's newly developed GraftAssure Combination Model (CM)-score mathematically combines the relative (percentage, or %) and absolute (copies per milliliter, or cp/mL) measurements of DNA fragments from the transplanted organ. In this study, the tight correlation between the novel GraftAssure CM-score and rejection indices suggests that the assay is reliably measuring complex biological signals in kidney transplantation.
Notably, GraftAssure’s CM-score outperformed both fractional and absolute measures on their own, in correlation analyses with the four indices of transplanted organ health, which are Antibody-mediated Rejection / Microvascular Inflammation (AMR/MVI index), T-Cell Mediated Rejection / Tubulointerstitial Inflammation (TCMR/TI index), Activity index, and Chronicity index. The rejection (AMR/MVI and TCMR/TI) indices were of particular interest to investigators because they are known to have a strong association with graft survival
As mentioned above, study authors also presented data at the World Transplant Congress in 2025 that showed an improvement in positive predictive value from around 50% seen in the literature to an unprecedented value of above 80% using the GraftAssure CM-score (at 25% prevalence, which is often seen in for-cause settings) enabling the rule-in option for transplanted organ rejection for the first time. That study is currently under review for publication.
“Kidney transplant rejection is increasingly understood as a biological continuum rather than a simple yes-or-no diagnosis,” said iMDx Chief Science Officer Prof. Dr. Ekkehard Schuetz, who co-authored the study. “Our results show that dd-cfDNA measured in the blood closely mirrors the inflammatory activity observed in transplant biopsies. One unexpected finding was that in TCMR, the total cell-free DNA increases with severity, which resulted in a loss of correlation for dd-cfDNA when reported as percentage. Whereas the Combination Model score showed the strongest correlation with TCMR. This supports the concept that integrated blood-based monitoring can provide clinicians with a real-time view of graft immune activity and help guide earlier intervention as rejection begins to develop.”
The study was investigator-initiated and investigator-led. Insight Molecular Diagnostics provided dd-cfDNA measurements used in the analysis.
References
| (1) | Vaulet T et al. Continuous indices to assess the phenotypic spectrum of kidney transplant rejection. Nat Commun. 2025;16(1):10417 |
iMDx Transplant Products and Product Candidates in Development
iMDx's flagship transplant testing technology quantifies a molecular biomarker known as donor-derived cell-free DNA (dd-cfDNA). The company's scientists in Germany and the U.S. have played a critical role over the past decade in developing the science that helped establish dd-cfDNA as a trusted biomarker of transplant rejection. iMDx is commercializing this technology using a market-disruptive business strategy. Under the GraftAssure
- GraftAssureCore - The company's laboratory-developed test (LDT), currently reimbursed by CMS and performed at iMDx's CLIA-certified laboratory in Nashville.
- GraftAssureIQ - A research-use-only (RUO) kit intended and labeled for non-clinical applications.
- GraftAssureDx - The in vitro diagnostic (IVD) kit currently in development for use in clinical decision-making.
About Insight Molecular Diagnostics, Inc.
Insight Molecular Diagnostics is a pioneering diagnostics technology company whose mission is to democratize access to novel molecular diagnostic testing to improve patient outcomes. Investors may visit https://investors.imdxinc.com/ for more information.
GraftAssureCore
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Shopify Makes a Major Move into AI-Powered Commerce: Could This Be the Next Driver of Growth?
Gig Economy Stocks Q4 Earnings Analysis: Uber (NYSE:UBER) Stands Out
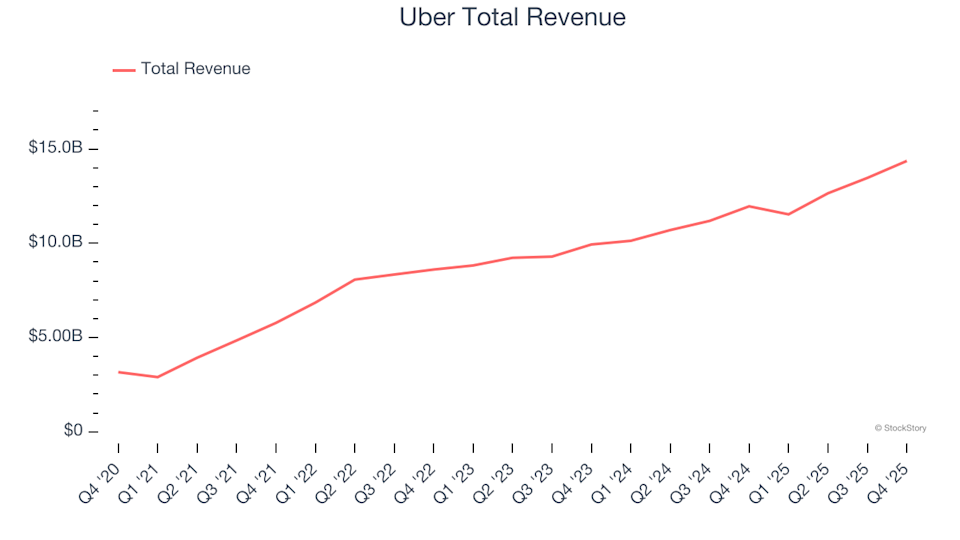
Gig Economy Stocks Q4 Earnings Analysis: Uber (NYSE:UBER) Stands Out

BoE: Energy shock complicates easing path – Nomura

