ImmunityBio Shares Surge 295% Year-to-Date: Should You Buy, Hold, or Sell?
ImmunityBio Shares Surge Dramatically in 2024
ImmunityBio (IBRX) has seen its stock price soar by nearly 300% since the start of the year, far surpassing the industry’s modest 6% increase. This remarkable performance has also outpaced both the broader healthcare sector and the S&P 500, as illustrated below.
IBRX Outpaces Peers and Major Indexes
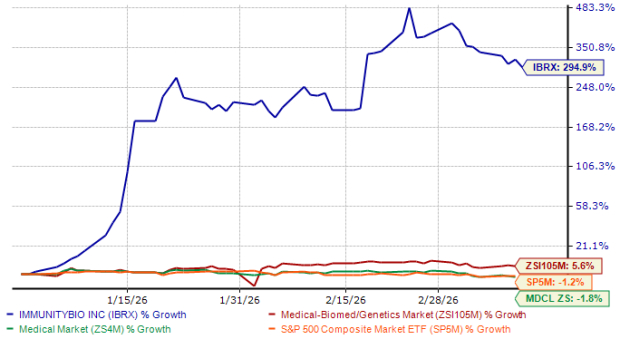
Source: Zacks Investment Research
This impressive rally is largely fueled by the strong commercial results of Anktiva, ImmunityBio’s flagship and only marketed therapy. Approved by the FDA for use alongside Bacillus Calmette-Guérin (BCG), Anktiva treats adults with BCG-unresponsive non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS), regardless of papillary tumor presence.
Let’s take a closer look at ImmunityBio’s core business to understand the factors behind its recent momentum.
Anktiva: The Main Engine Behind ImmunityBio’s Revenue Growth
Since its regulatory approval in 2024, Anktiva’s market introduction has been notably successful. In the most recent financial update, ImmunityBio reported robust results for both the fourth quarter and the full year of 2025, with annual net product sales reaching $113 million—a staggering 700% increase from the previous year. The company attributes much of this growth to repeat prescriptions, indicating that healthcare providers are increasingly confident in the drug’s safety and effectiveness.
Another factor driving demand is the ongoing shortage of BCG, which has complicated bladder cancer treatment. Because Anktiva is administered with BCG, physicians are prioritizing this combination for eligible patients, further boosting sales.
ImmunityBio is actively seeking to broaden Anktiva’s approved uses. The company recently resubmitted a supplemental application to the FDA, aiming to expand the drug’s label for use in BCG-unresponsive NMIBC with papillary disease. This submission, which follows extensive discussions with regulators and includes new clinical data, could pave the way for more patients to benefit from Anktiva if approved.
Internationally, ImmunityBio is making significant strides. Since early 2026, Anktiva has gained regulatory clearance for BCG-unresponsive NMIBC in both the European Union and Saudi Arabia. These milestones are expected to accelerate the company’s global commercialization efforts and open up new revenue opportunities as ImmunityBio establishes partnerships and distribution channels abroad.
Significantly, ImmunityBio is also moving beyond bladder cancer. In January, Anktiva received its first approval for non-small cell lung cancer (NSCLC) in Saudi Arabia, marking its entry into a major oncology market. The company plans to engage with the FDA later this year to pursue similar approval for NSCLC in the United States, which could further expand Anktiva’s commercial reach.
Promising Clinical Pipeline for Anktiva Expansion
In addition to its commercial success, ImmunityBio is advancing a pipeline of clinical programs designed to extend Anktiva’s benefits to a wider range of cancers.
The company is conducting multiple clinical trials across both solid tumors and blood cancers as part of its Cancer BioShield platform. Anktiva serves as a foundational immunotherapy in these studies, aiming to activate immune cells like natural killer (NK) cells and T cells.
Within bladder cancer, a randomized trial is underway to evaluate Anktiva plus BCG in patients who have not previously received BCG, with a potential FDA submission planned for later this year. Additional research is exploring the drug’s effectiveness in other bladder cancer settings, potentially increasing the number of patients who could benefit.
ImmunityBio is also investigating Anktiva in combination with standard treatments or CAR-NK cell therapies for several challenging cancers, including NSCLC, pancreatic cancer, glioblastoma, colorectal cancer, and hepatocellular carcinoma.
If these clinical trials yield positive results, Anktiva could become a cornerstone immunotherapy for multiple cancer types, supporting further regulatory submissions and long-term business growth.
Facing Stiff Competition in Oncology
Despite its rapid progress, ImmunityBio operates in a fiercely competitive oncology market dominated by major pharmaceutical companies. Anktiva competes with established immunotherapies such as Merck’s Keytruda, Bristol Myers Squibb’s Opdivo, and Roche’s Tecentriq, all of which are widely used across various cancer indications targeted by ImmunityBio.
These industry giants not only have approved therapies but also benefit from vast financial resources, global commercial operations, and robust supply chains. Their deep experience and strong relationships with healthcare providers present significant hurdles for newer entrants like ImmunityBio.
Risks of Relying on a Single Product
A major vulnerability for ImmunityBio is its dependence on Anktiva as its sole commercial product and primary revenue source. Any decline in demand, regulatory delays, or unforeseen safety issues could have a significant negative impact on the company’s financial health.
Valuation and Analyst Expectations for IBRX
ImmunityBio’s stock is currently valued at a premium compared to its peers. The forward price-to-sales (P/S) ratio stands at 32.59, well above the industry average of 2.00.
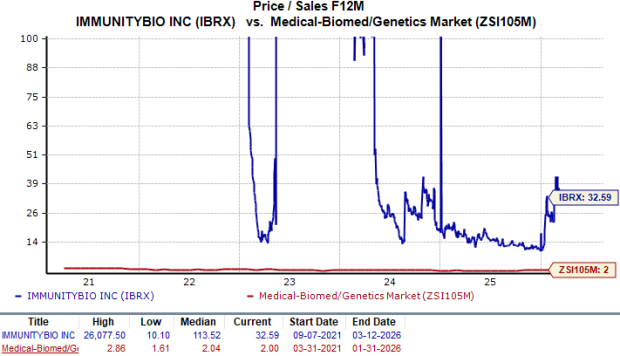
Source: Zacks Investment Research
Analyst estimates for ImmunityBio’s earnings per share (EPS) in 2026 and 2027 have shown mixed trends over the past month.
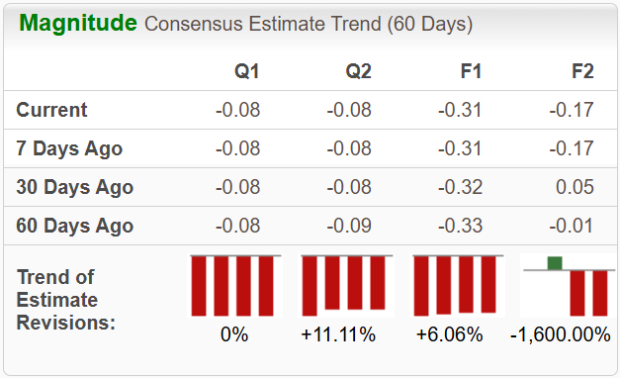
Source: Zacks Investment Research
Investment Outlook: What’s Next for IBRX?
Despite facing competition from industry heavyweights, Anktiva’s early commercial success demonstrates growing market acceptance. Strong initial sales and expanding regulatory approvals suggest that the drug could continue to drive ImmunityBio’s revenue growth in the years ahead.
Although the stock trades at a higher valuation than its industry peers, investors may consider holding shares of this Zacks Rank #3 (Hold) company due to its growth prospects.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Ulta Beauty’s Explosive Start to the Year Has Stalled. Here’s Why.
NuScale Power Shares Drop Close to 20% at the Beginning of 2026

