IBRX Stock: Anktiva Receives Approvals and Future Steps Ahead
ImmunityBio Enters Commercial Stage with Anktiva at the Core
ImmunityBio has transitioned into a commercial-stage immunotherapy company, with its immediate future closely linked to the performance of its flagship product, Anktiva. This therapy now serves as the foundation for both the company's revenue and its upcoming regulatory and market milestones.
As demand in the United States rises, international approvals are secured, and efforts to broaden Anktiva’s indications progress, the importance of operational execution and regulatory developments will be as significant as clinical advancements by 2026.
Anktiva: The Driving Force Behind ImmunityBio’s Business
ImmunityBio’s strategy centers on immunotherapies that stimulate both innate and adaptive immune responses, with Anktiva leading its commercial expansion. The company’s current income is largely derived from Anktiva sales, making its market performance pivotal for investors.
Anktiva is an interleukin-15 receptor superagonist antibody-cytokine fusion protein, designed to activate natural killer cells, cytotoxic T cells, and memory T cells. This mechanism supports its role as a versatile immuno-oncology platform, with the potential for broader applications in the future.
In the U.S., Anktiva is approved for use with bacillus Calmette-Guérin (BCG) in adults who have BCG-unresponsive non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS), regardless of the presence of papillary tumors. This indication forms the basis of the company’s current commercial activities and highlights the initial expansion opportunities being pursued.
Revenue Surge in 2025: Key Factors
In 2025, ImmunityBio reported total revenues of $113.3 million, a significant increase from $14.7 million in 2024. Nearly all of this, $113.0 million, came from product sales, reflecting the first full year of Anktiva’s commercial rollout.
This growth is directly linked to the product’s launch timeline. Following FDA approval in April 2024, shipments began in May, paving the way for broader adoption and higher revenues throughout 2025.
Other income sources, such as bioreactor sales and nonexclusive licensing, contributed only $0.3 million and remain non-essential to the company’s financial outlook. Essentially, ImmunityBio’s growth is currently fueled by a single commercial product rather than a diversified portfolio.
U.S. Market Momentum Heading into 2026
Trends at the end of 2025 were particularly notable. Net product sales in the fourth quarter reached $38.3 million, with management noting a 20% increase from the previous quarter—evidence of growing demand as the company entered 2026.
Additionally, ImmunityBio pointed to increasing repeat prescriptions from physicians, a positive sign for sustained adoption in the bladder cancer market as providers become more familiar with the therapy.
Reimbursement processes also improved, with the introduction of a permanent J-code on January 1, 2025, simplifying payment procedures and supporting wider uptake—an important factor for early-stage oncology launches.
ImmunityBio, Inc. Price and Consensus
Global Expansion: New Markets in 2026
ImmunityBio’s international reach grew significantly as 2026 approached. The European Commission granted conditional marketing authorization for Anktiva in February 2026 for the same primary indication, complementing earlier approval in the United Kingdom and paving the way for further commercial expansion outside the U.S.
Saudi Arabia also approved Anktiva for BCG-unresponsive NMIBC and conditionally for use with checkpoint inhibitors in metastatic non-small cell lung cancer (NSCLC), broadening the company’s geographic and therapeutic footprint.
In Europe, partner Accord Healthcare plans to launch Anktiva across 31 countries, focusing first on the largest markets, with Germany slated for an early rollout in 2026. ImmunityBio has indicated that there are no supply issues for these initial launches, which is crucial as reimbursement and distribution are managed on a country-by-country basis.
Key Label Expansion Opportunities
Expanding Anktiva’s approved uses remains a major growth driver, especially within non-muscle invasive bladder cancer. In phase II/III trials for BCG-unresponsive CIS, Anktiva combined with BCG achieved a 71% complete response rate, with durable results extending beyond 53 months and high rates of bladder preservation.
The company is also targeting earlier treatment settings. Interim results from a mid-stage study in BCG-naïve NMIBC CIS showed improved 6- and 9-month complete response rates for Anktiva plus BCG. ImmunityBio aims to submit for U.S. approval based on this data before the end of 2026.
In the near term, a supplemental biologics license application was resubmitted on March 9, seeking to expand Anktiva’s label for use with BCG in BCG-unresponsive NMIBC with papillary disease, following additional FDA information requests.
Risks: Product Concentration and Competitive Landscape
The company’s reliance on Anktiva presents a significant risk; any setbacks in development or regulatory approval could have a major impact on growth prospects.
Competition is also intense. ImmunityBio faces established players in the immuno-oncology space, particularly in metastatic NSCLC, where combination therapies with checkpoint inhibitors compete with well-known treatments like Merck’s Keytruda and Bristol Myers Squibb’s Opdivo, both of which are leading PD-1 checkpoint inhibitors.
Looking ahead to 2026 and 2027, the company’s success will depend on maintaining U.S. momentum, converting European approvals into successful launches, and advancing regulatory filings that could broaden Anktiva’s indications and diversify revenue streams.
Current Zacks Rank for IBRX
ImmunityBio currently holds a Zacks Rank #3 (Hold).
Newly Released: Zacks Top 10 Stocks for 2026
There’s still time to get early access to Zacks’ top 10 stock picks for 2026. Curated by Zacks Director of Research Sheraz Mian, this portfolio has delivered impressive and consistent results.
From its inception in 2012 through November 2025, the Zacks Top 10 Stocks portfolio achieved gains of +2,530.8%, far outpacing the S&P 500’s +570.3% return.
Sheraz has reviewed 4,400 companies covered by the Zacks Rank and selected the 10 best stocks to buy and hold for 2026. You can be among the first to access these high-potential picks.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Pangaea, Curtiss-Wright, Hexcel, Ameresco, and Mercury Systems Stocks Are Declining—Here’s What’s Important
Established Link Between Ripple (XRP) and Two Largest Credit Card Issuers
TRUMP (OfficialTrump) 24-hour volatility at 55.8%: Mar-a-Lago top holder invitation event drives surge
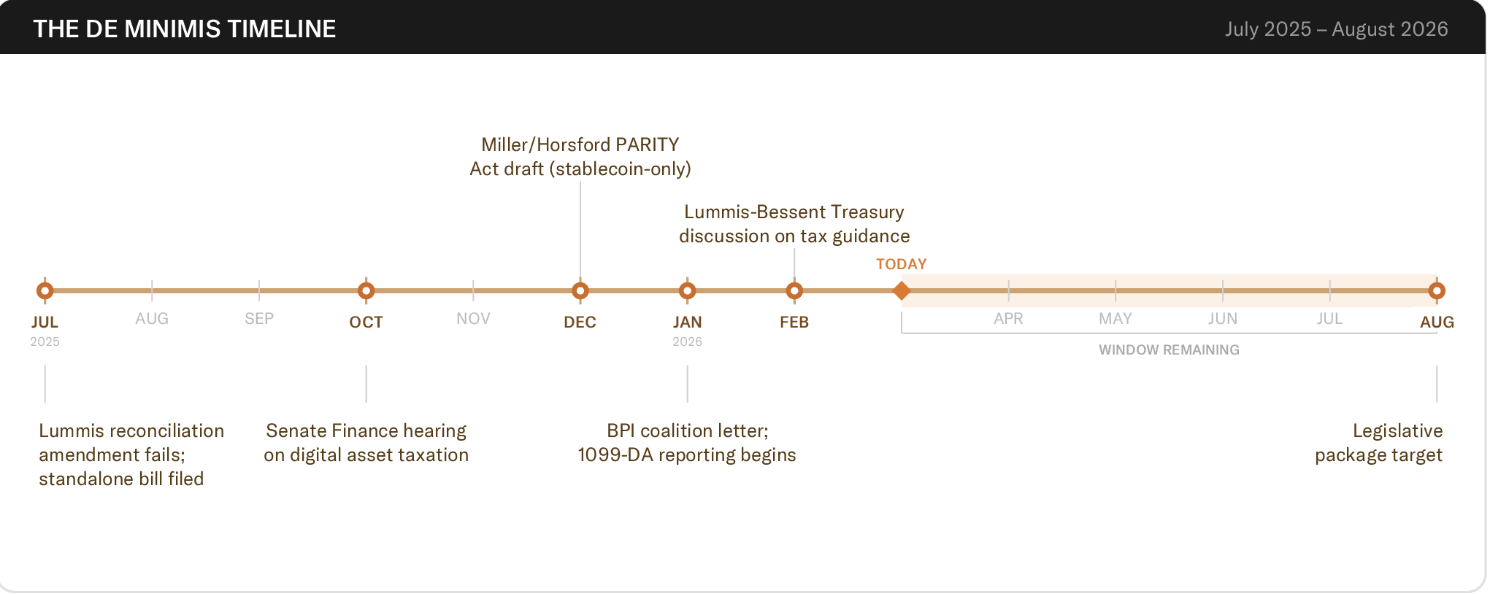
BPI targets August for BTC tax relief, but warns time is running out