FDA Approval of Abbott's CardioMEMS Hero Drives $1.54 Billion Surge in Trading Activity Stock Ranks 103rd in Daily Volume
Market Snapshot
Abbott Laboratories (ABT) saw a notable surge in trading activity on February 27, 2026, with a volume of $1.54 billion, marking a 68.92% increase from the previous day. The stock ranked 103rd in terms of trading activity for the day, reflecting heightened investor interest. Despite the robust volume, the stock’s price rose modestly by 0.08%, closing at $120.45. The mixed performance suggests that while market participants engaged heavily with the stock, broader sentiment remained cautious, potentially due to the absence of significant post-approval price volatility following the recent FDA clearance for its CardioMEMS Hero device.
Key Drivers
The FDA’s approval of Abbott’s CardioMEMS Hero pulmonary artery pressure reader on February 27 emerged as the primary catalyst for the stock’s activity. This next-generation device, designed to remotely monitor heart failure patients, introduces several enhancements over its predecessor, including a 60% lighter form factor, Wi-Fi and cellular connectivity, and a streamlined design akin to a laptop case. These improvements address key usability challenges, such as portability and ease of daily readings, which could drive adoption among the 6.7 million U.S. heart failure patients. The device’s ability to transmit real-time pulmonary artery pressure data to clinicians enables early intervention, potentially reducing hospitalization rates by 57%, a metric that underscores its clinical and economic value.
The expanded indication for CardioMEMS, granted in February 2022, further amplifies the device’s market potential. By extending eligibility to an additional 1.2 million patients, AbbottABT-- has positioned itself to capture a larger share of the heart failure management market, which is projected to grow as the aging population increases. The commercial rollout of CardioMEMS Hero in the U.S. is expected to accelerate revenue growth, particularly as healthcare systems prioritize cost-saving technologies that mitigate hospital readmissions. Analysts have highlighted that the device’s integration into standard care protocols could drive long-term recurring revenue streams, as patients require ongoing monitoring and device replacements.
The strategic importance of CardioMEMS Hero is further underscored by Abbott’s broader innovation pipeline. The company’s commitment to advancing remote patient monitoring aligns with industry trends toward decentralized healthcare models, which have gained traction post-pandemic. The device’s design, which allows for consistent reading positions and virtual check-ins, addresses gaps in patient compliance and clinician accessibility. By enabling proactive management of heart failure progression, CardioMEMS Hero not only improves patient outcomes but also strengthens Abbott’s competitive positioning against rivals in the medical device sector.
While the stock’s 0.08% gain appears muted, the surge in trading volume indicates that investors are factoring in the long-term implications of the FDA approval. The market’s measured response may reflect anticipation of future catalysts, such as commercial launch timelines and early adoption rates. Additionally, Abbott’s recent $20 billion debt issuance to fund its Exact Sciences acquisition and the Novo Nordisk India partnership for a diabetes drug could influence investor sentiment in subsequent quarters. However, these developments are not directly tied to the CardioMEMS Hero announcement and are likely to be analyzed separately in the coming months.
In summary, the FDA clearance of CardioMEMS Hero represents a pivotal milestone for Abbott, combining technological innovation with unmet clinical needs. The device’s potential to reduce hospitalizations, coupled with its user-centric design, positions it as a key growth driver. While the immediate stock reaction was tempered, the underlying fundamentals suggest that the market will continue to monitor Abbott’s execution in scaling the CardioMEMS HF system and leveraging its remote monitoring capabilities to expand its footprint in chronic disease management.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
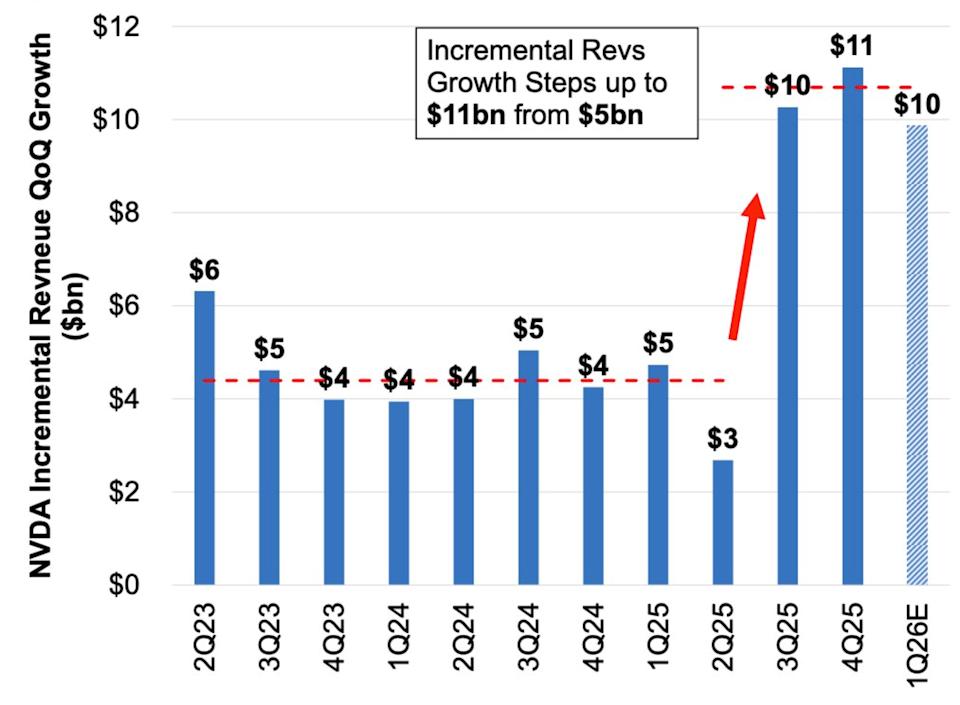
Here is the intriguing puzzle behind Nvidia's stock
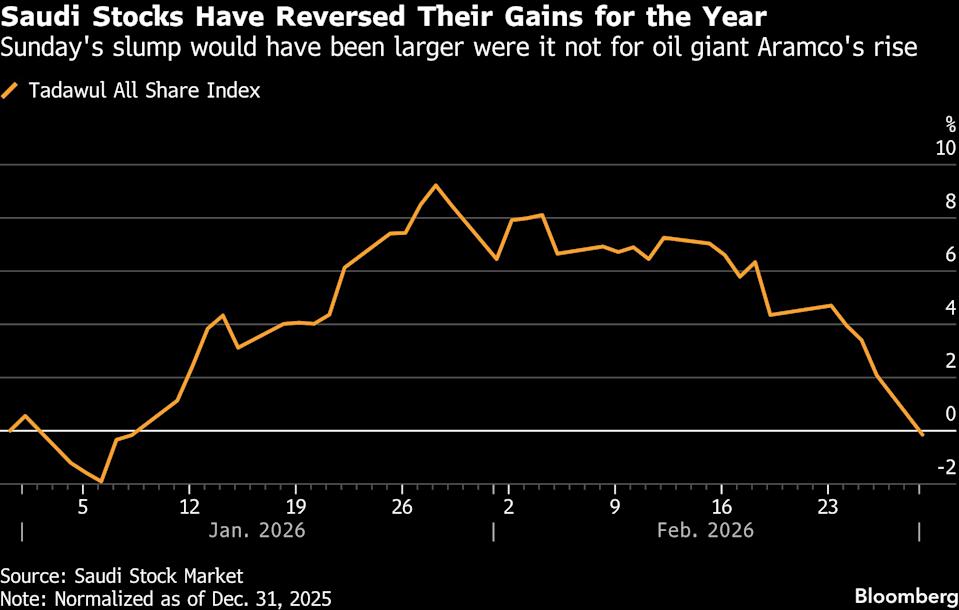
Saudi and Egyptian stock exchanges tumble amid regional turmoil caused by US-Iran tensions
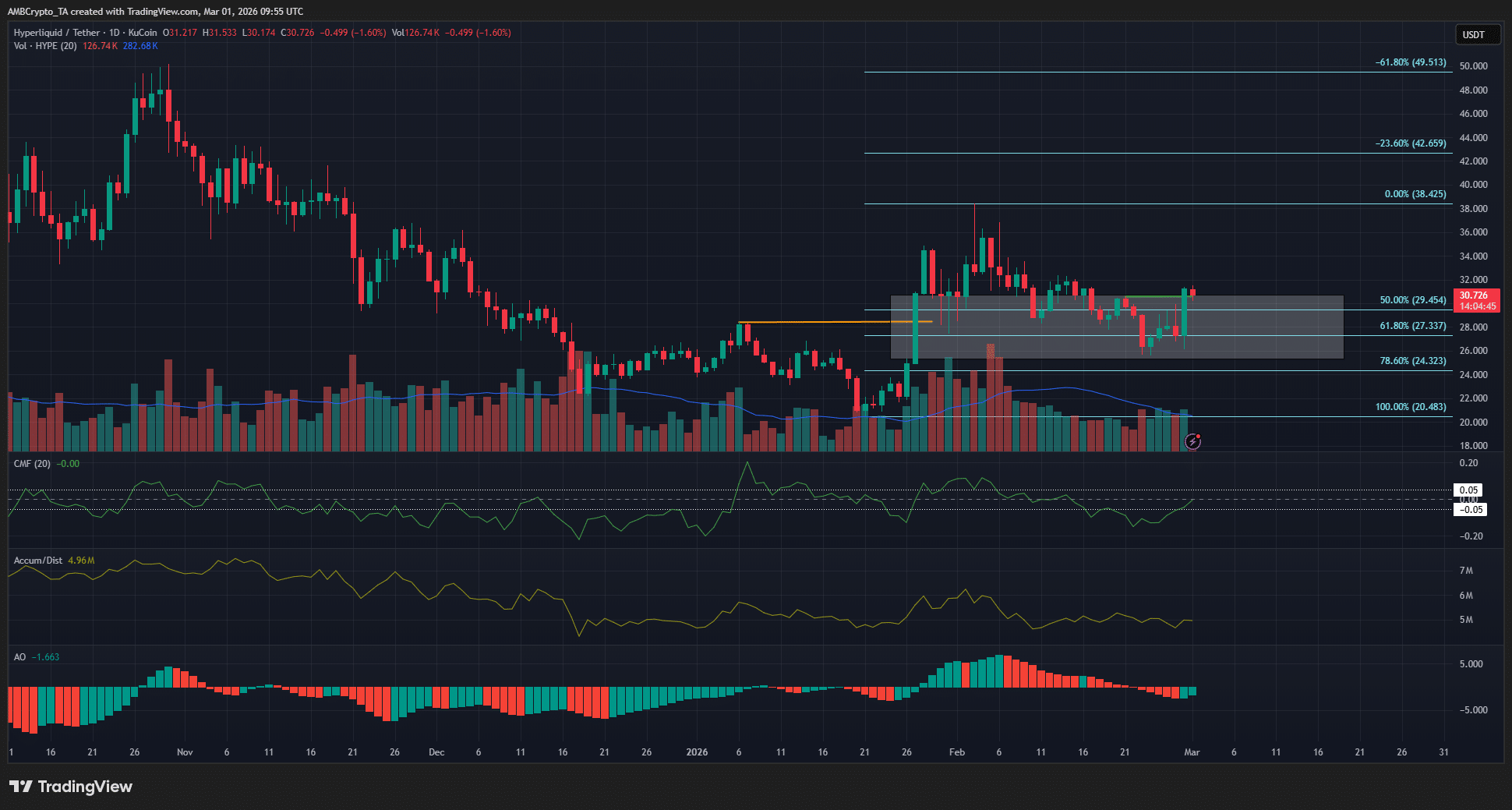
Hyperliquid price prediction – HYPE eyes $38, but watch THIS golden pocket first
If You’re Bearish on XRP, Listen to What David Schwartz Said
