FDA’s Top Vaccine Official Vinay Prasad Will Step Down From His Role Next Month
FDA Vaccine and Gene Therapy Chief to Step Down

Image credit: US Food and Drug Administration
The director overseeing vaccines and gene therapies at the US Food and Drug Administration is set to leave the agency after a period marked by controversy, a move that sparked a surge in biotech stock prices late Friday.
Vinay Prasad, who leads the Center for Biologics Evaluation and Research, will exit his role next month to resume his academic career. Prasad developed a reputation for occasionally overruling his scientific team and for his confrontational leadership style, which some critics argued hindered scientific progress and delayed new drug approvals.
Top Stories from Bloomberg
News of Prasad’s resignation triggered a rally in certain biotech stocks. UniQure NV’s shares soared by 55% in after-hours trading. The company has been publicly at odds with Prasad’s division regarding the approval process for its Huntington’s disease gene therapy. Regenxbio Inc. also saw its shares climb 16% after the FDA declined to approve its gene therapy for Hunter syndrome last month.
Prasad began his tenure at the FDA in May 2025. He briefly left in late July following criticism from conservative commentators over his management of Sarepta Therapeutics Inc.’s gene therapy, but returned weeks later with the backing of FDA Commissioner Marty Makary.
Commissioner Makary expressed gratitude for Prasad’s contributions in a post on X, highlighting that the division achieved a record number of approvals in December. Makary indicated that a replacement will be named before Prasad’s departure.
While at the FDA, Prasad launched several initiatives aimed at accelerating the approval of treatments for rare diseases. However, some biotech firms have complained that regulatory requirements often changed unexpectedly, complicating the approval process for new therapies.
Previously a professor of epidemiology at the University of California, San Francisco, Prasad became known for his criticism of Covid vaccines for children and for challenging companies that do not use placebo-controlled trials for their products.
Prasad succeeded Peter Marks, who was removed from the position a year earlier after a dispute with HHS Secretary Robert F. Kennedy Jr. regarding vaccine policy.
The Wall Street Journal was the first to report Prasad’s planned exit.
Reporting assistance by Nyah Phengsitthy and John Tozzi.
More from Bloomberg Businessweek
©2026 Bloomberg L.P.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
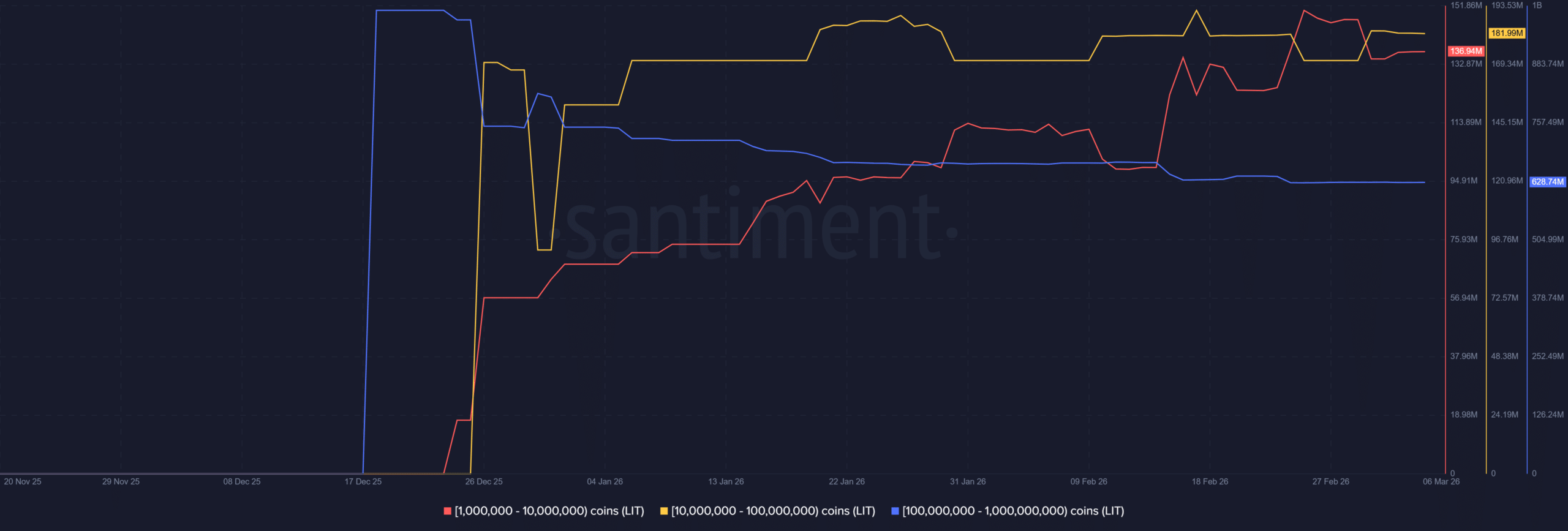
Liquidity shock? LIT drops 16% after Justin Sun pulls funds from Lighter
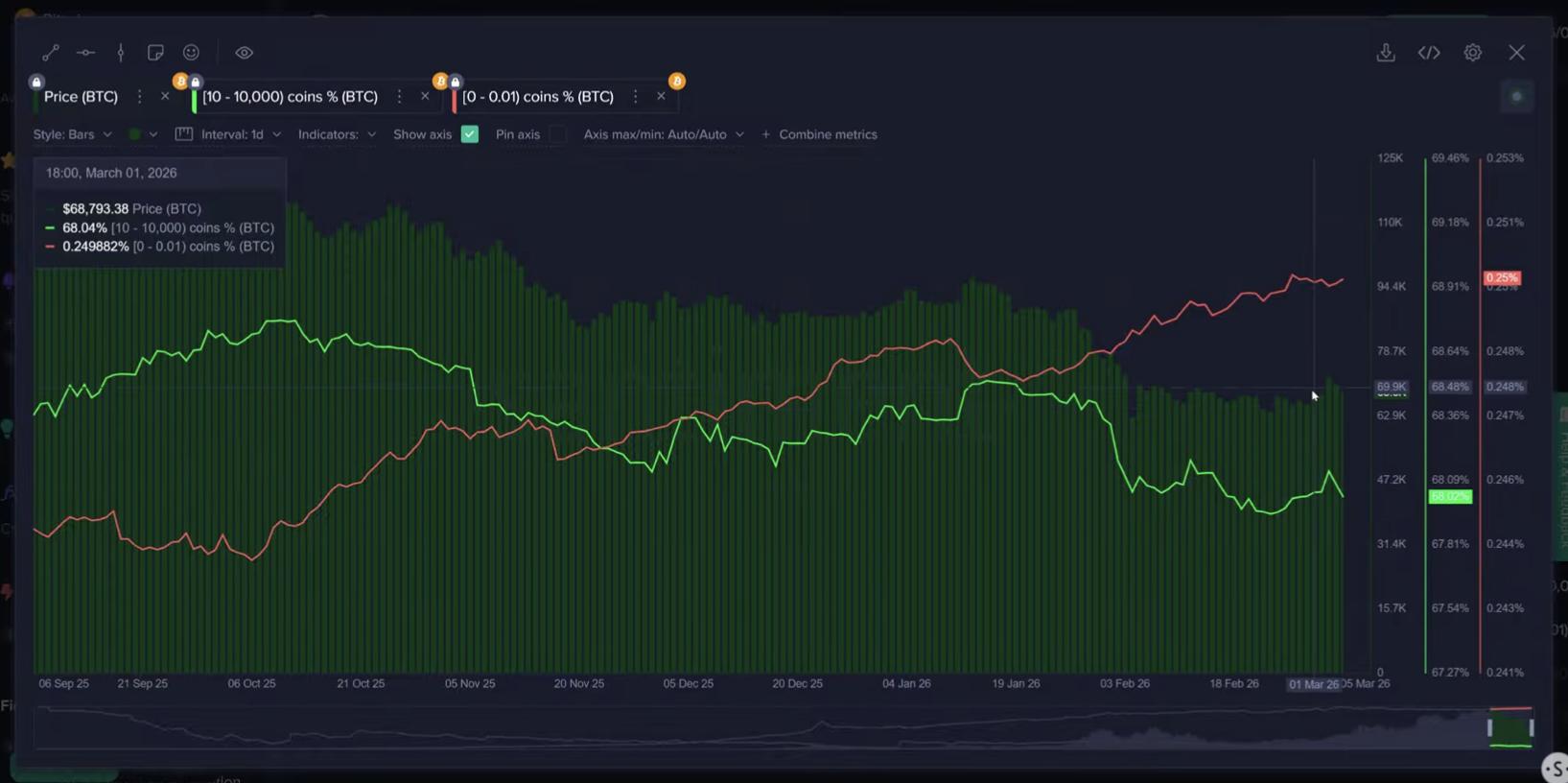
Bitcoin dip may not be over as retail ramps up buying below $70K: Santiment
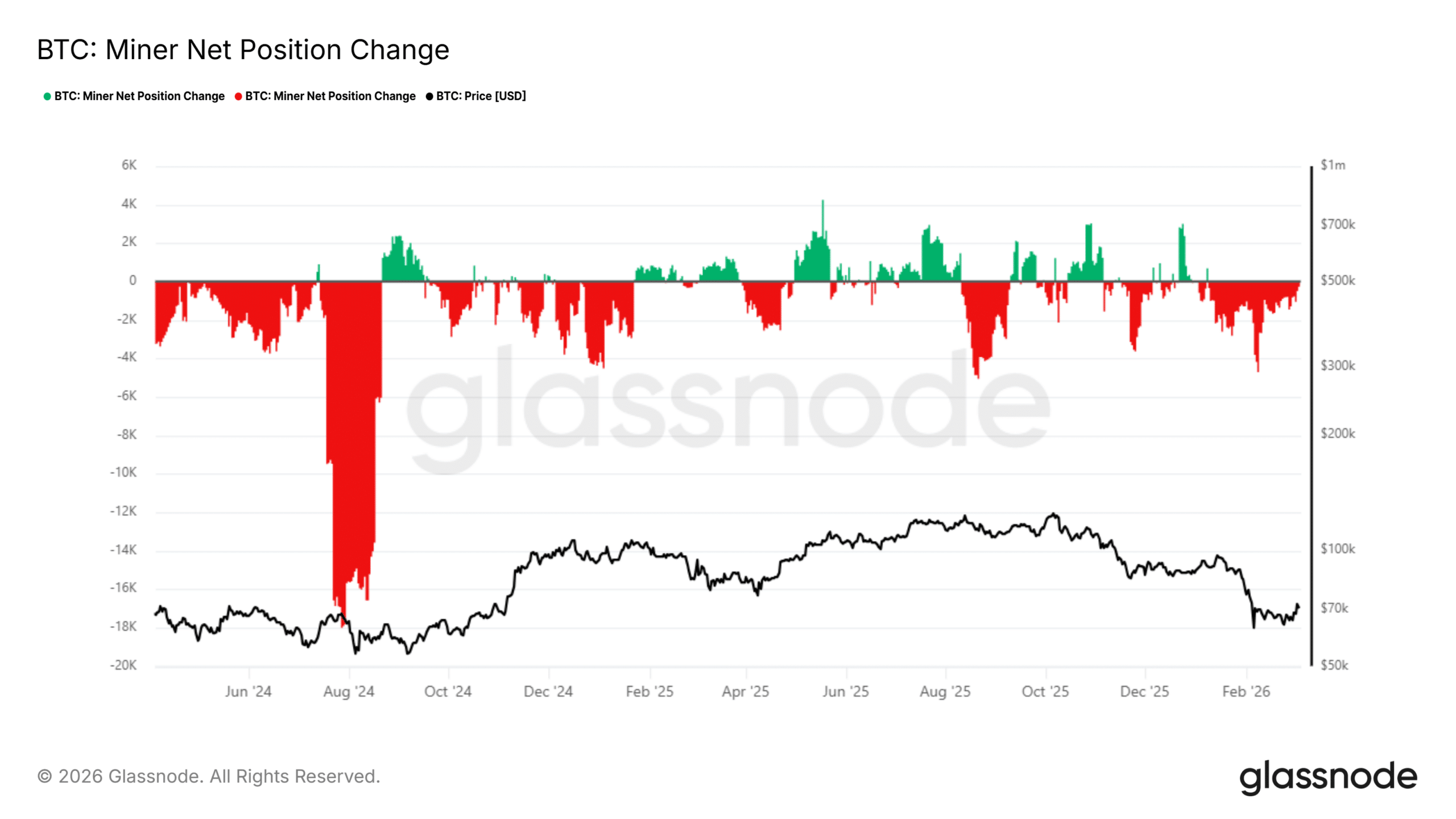
CleanSpark and Bitcoin miners’ selling spree – Is the miner HODL era ending?