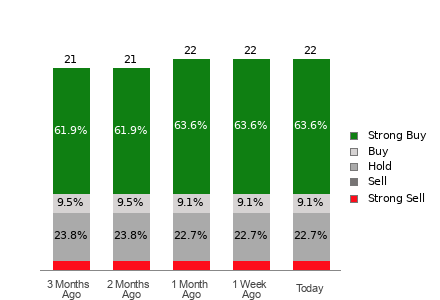
Femasys Advances FemBloc Toward U.S. Approval with Initiation of Pivotal Trial Enrollment
-- Enrollment begins in the FDA-authorized FINALE pivotal trial evaluating FemBloc, a first-of-its-kind non-surgical permanent birth control, following positive clinical data --
ATLANTA, March 10, 2026 (GLOBE NEWSWIRE) -- Femasys Inc. (NASDAQ: FEMY), a leading biomedical innovator focused on making fertility and non-surgical permanent birth control more accessible and cost-effective for women worldwide, announced today the initiation of patient enrollment in the FINALE pivotal clinical trial evaluating FemBloc. This milestone follows U.S. Food and Drug Administration (FDA) Investigational Device Exemption (IDE) approval to advance the study based on positive clinical data generated in its earlier phase. FemBloc has already received regulatory approvals and is being commercialized in Europe and other select countries outside the United States as a non-surgical alternative to the surgical procedures currently used for permanent birth control. Completion of the FINALE study is expected to support a future U.S. regulatory submission.
“Initiating patient enrollment in the final phase of the FINALE trial marks an important milestone toward unlocking the U.S. market for FemBloc and reflects our continued commitment to advancing women’s healthcare,” said Kathy Lee-Sepsick, Chief Executive Officer and Founder of Femasys Inc. “Following years of development, clinical validation, and regulatory progress, including approvals outside the United States, this pivotal trial represents the final step in executing our U.S. strategy and advancing long-term value creation for Femasys stakeholders.”
“Having been involved in the FemBloc clinical program from its early studies through this pivotal phase, I am pleased to see the FINALE trial advance to its final stage,” said Dr. Scott Chudnoff, Principal Investigator of the FINALE trial, Chair of Obstetrics & Gynecology at Maimonides Medical Center. “A non-surgical approach has the potential to expand access while reducing the risks associated with surgery, addressing a longstanding unmet need for women in the U.S.”
Permanent birth control is one of the most widely used contraceptive methods worldwide, yet current options require surgery, anesthesia, abdominal incisions, and recovery time, introducing procedural risks and increasing healthcare costs. FemBloc is a first-of-its-kind, non-surgical solution designed to address this unmet need in women’s reproductive health. Using a patented delivery system, FemBloc places a proprietary blended polymer into both fallopian tubes, where it safely degrades and forms natural scar tissue for permanent occlusion. By eliminating the complexities associated with surgical sterilization, FemBloc has the potential to expand access to permanent contraception while reducing the burden on healthcare systems. Millions of women worldwide undergo surgical procedures for permanent birth control each year, highlighting the need for a non-surgical alternative.
About the FINALE Pivotal Clinical Trial
The FINALE (Prospective Multi-Center Trial for FemBloc INtratubal Occlusion for TranscervicAL PErmanent Birth Control) trial is a prospective, multi-center, open-label, single-arm pivotal study evaluating the safety and effectiveness of FemBloc. An initial roll-in enrollment of 50 women was completed to assess preliminary safety data, together with data from prior IDE-approved studies, after which the U.S. Food and Drug Administration (FDA) authorized continuation of the trial to enroll the remaining participants. The primary endpoint is pregnancy rate, assessed after 401 women have used FemBloc for one year, with an interim analysis planned after 300 participants complete one year of use. Participants will be followed annually for five years post-procedure. For more information, visit .
About Femasys
Femasys is a leading biomedical innovator focused on making fertility and non-surgical permanent birth control more accessible and cost-effective for women worldwide through its broad, patent-protected portfolio of novel, in-office therapeutic and diagnostic products. As a U.S. manufacturer with global regulatory approvals, Femasys is actively commercializing its lead product innovations in the U.S. and key international markets. Femasys’ fertility portfolio includes FemaSeed
FemBloc
References
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Down 16.6% in 4 Weeks, Here's Why You Should You Buy the Dip in LCI (LCII)
3 Financials Get Fresh Buy Ratings: Wall Street Turns Bullish on UWMC, RKT and SLRC
STMicroelectronics (STM) Jumps 6.9%: Does the Stock Have More Room to Grow?
XPeng (XPEV) Viewed Favorably by Analysts: Does the Claim Hold Up?