News
Stay up to date on the latest crypto trends with our expert, in-depth coverage.

1Bitget UEX Daily | Trump Pledges to Safeguard Crude Oil Transport; Oil Prices Surge and Pull Back; Gold and Silver Plunge as Dollar Strengthens (March 04, 2026)2Locals prefer satoshis to dollars, says Africa Bitcoin chair Stafford Masie3'No longer a choice': Bitwise CIO says US-Iran strikes put crypto in primary market role
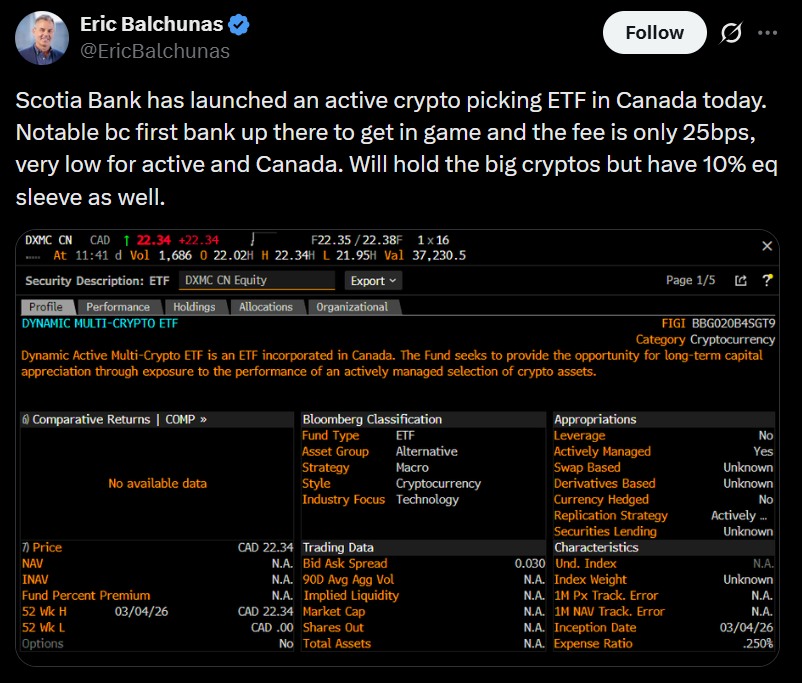
Canada's Scotiabank launches multi-crypto ETF with 3iQ
Cointelegraph·2026/03/04 21:03

Dogecoin Pumps as Bitcoin Pops, Reversing Recent DOGE Losing Streak
Decrypt·2026/03/04 20:53

Bitcoin Treasury Inflows Decelerate: Company Holdings Compared to ETF Withdrawals
101 finance·2026/03/04 20:45

Google reaches agreement with Epic Games, reduces Play Store commission fees to 20%
101 finance·2026/03/04 20:36
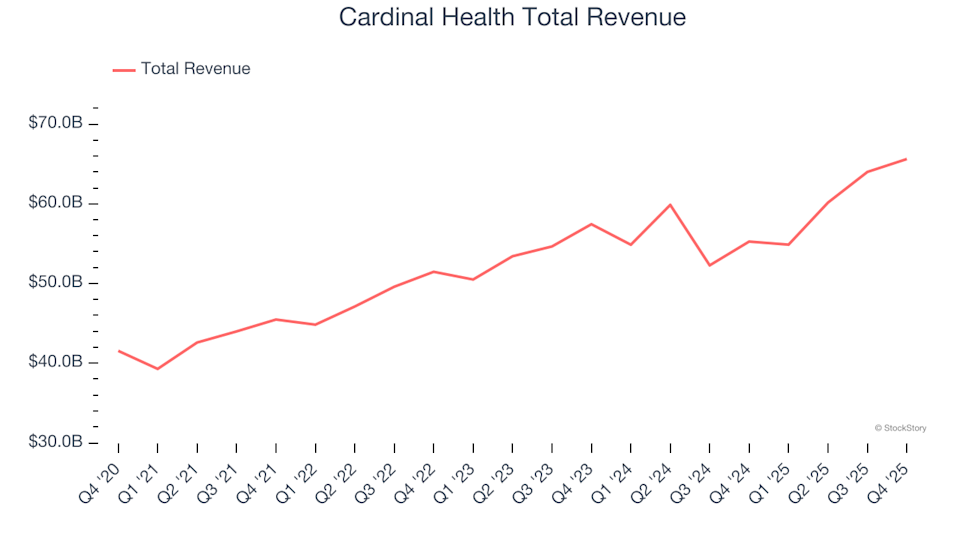
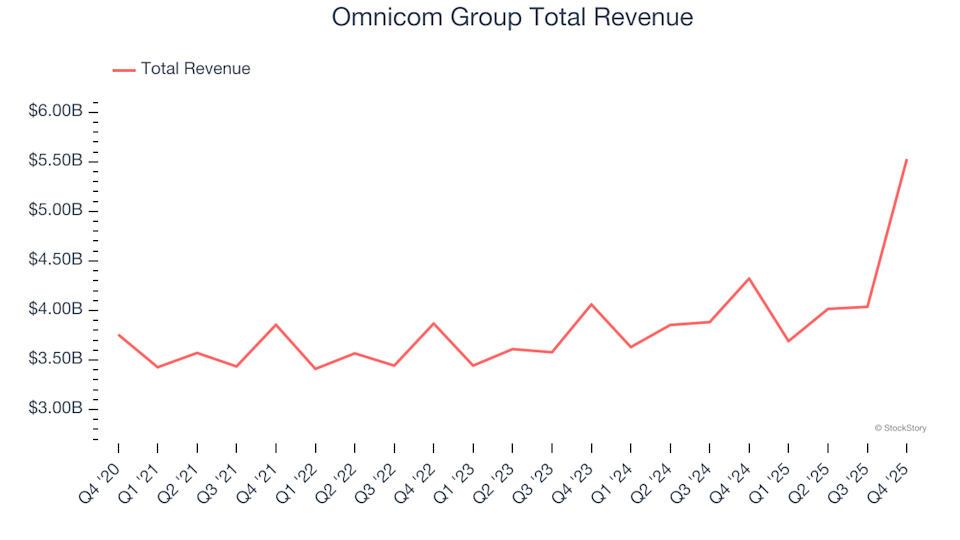
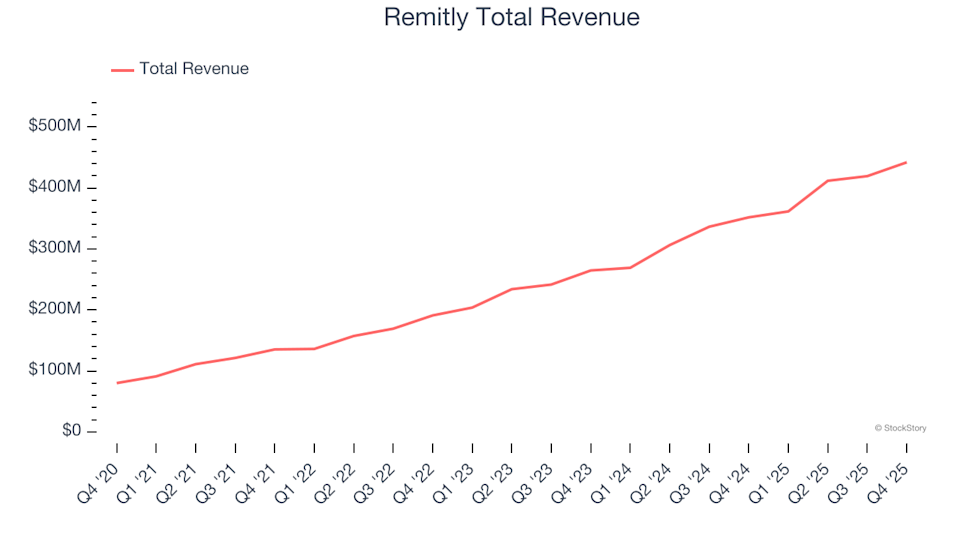
Reflecting on the fourth quarter earnings of financial technology stocks: Remitly (NASDAQ:RELY)
101 finance·2026/03/04 20:30

Reflecting on the fourth quarter earnings of financial technology stocks: Remitly (NASDAQ:RELY)
101 finance·2026/03/04 20:30
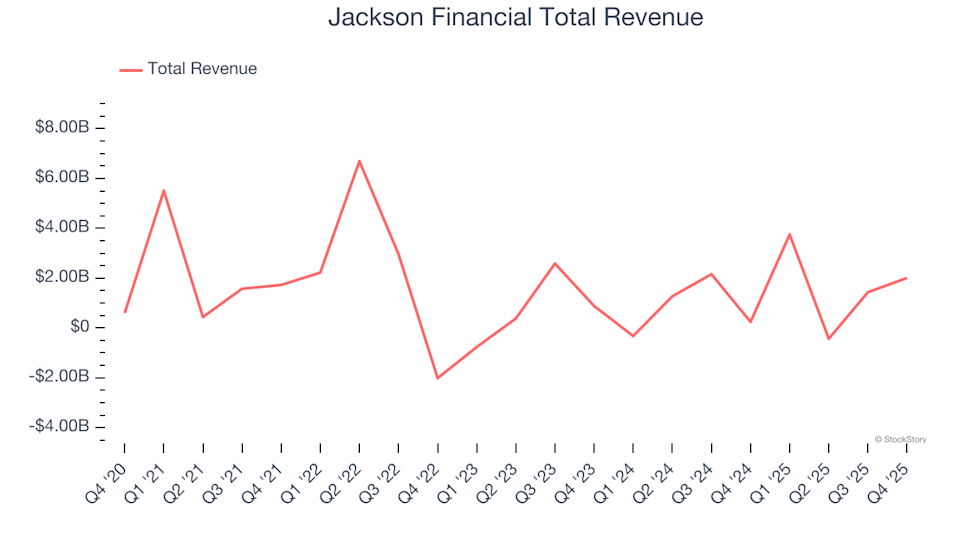
Firing on All Cylinders: Jackson Financial (NYSE:JXN) Sets the Pace with Q4 Earnings
101 finance·2026/03/04 20:30

Diamond Hill’s Fourth Quarter Success: Examining the Disparity Between Results and Expectations
101 finance·2026/03/04 20:24
Flash
21:12
Biotechnology company PepGen Inc. recently reaffirmed its clinical development timeline, clearly stating that it will release research data from the 5 mg/kg dose group in the Freedom-2 clinical trial in the first quarter of 2026.The reiteration of this timeline provides clear expectations for investors focused on the treatment of neuromuscular diseases. In addition, for the higher-dose 10 mg/kg cohort, the company plans to announce relevant clinical results in the second half of 2026. The sequential disclosure of these two key data sets will gradually reveal the safety and efficacy of its candidate therapy in patients with Duchenne muscular dystrophy. The maintenance of this guidance reflects the company’s strict control over the clinical development process, and also provides an important time marker for the market to assess the value of PepGen Inc.’s technology platform and drug development potential.
21:11
Users of a certain exchange may experience transaction delays or system performance degradation when conducting peer-to-peer transfers on mobile devices.The company stated that it is closely monitoring the relevant technical issues and will release a solution as soon as possible.
21:10
PepGen Inc. has received regulatory approval to launch its Freedom2 clinical trial program in South Korea, Australia, and New Zealand.This development marks an important step for the company in expanding its global clinical development footprint, making it possible for patients in relevant regions to participate in innovative therapy research.
News